Advertisements
Advertisements
Question
What happens when ethyl chloride is treated with aqueous KOH?
Advertisements
Solution 1
\[\ce{CH3 - CH2 - Cl ->[KOH/H2O]\underset{(S_N2 reaction)}{CH3 - CH2 - OH}}\]
Solution 2
When ethyl chloride is treated with aqueous KOH, it undergoes dehydrohalogenation, which eliminates hydrogen and halogen atoms from nearby carbon atoms, resulting in the production of an alkene.
\[\ce{\underset{Ethyl chloride}{CH3 - CH2 - Cl} + KOH (aq) ->[hydrolysis][\Delta] \underset{Ethyl alcohol}{CH3 - CH2 - OH} + KCl}\]
RELATED QUESTIONS
Write the isomers of the compound having the formula C4H9Br.
What are ambident nucleophiles? Explain with an example.
Arrange the compounds of the following set in order of reactivity towards SN2 displacement:
1-Bromobutane, 1-Bromo-2, 2-dimethylpropane, 1-Bromo-2-methylbutane, 1-Bromo-3-methylbutane
Tertiary alkyl halides are practically inert to substitution by SN2 mechanism because of ____________.
An important chemical method to resolve a racemic mixture makes use of the formation of ______.
Which of the following compounds is optically active?
SN1 reaction of alkyl halides lead to ___________.
The increasing order of reactivity towards SN1 mechanism is:
(I) \[\begin{array}{cc}
\ce{CH3-CH-CH2-CH3}\\
|\phantom{........}\\
\ce{CH3}\phantom{.....}
\end{array}\]
(II) CH3CH2CH2Cl
(III) P–CH3O–C6H4–CH2Cl
Assertion: KCN reacts with methyl chloride to give methyl isocyanide.
Reason: CN– is an ambident nucleophile.

Which of the statements are correct about above reaction?
(i) (a) and (e) both are nucleophiles.
(ii) In (c) carbon atom is sp3 hybridised.
(iii) In (c) carbon atom is sp2 hybridised.
(iv) (a) and (e) both are electrophiles.
Write the structures and names of the compounds formed when compound ‘A’ with molecular formula, \[\ce{C7H8}\] is treated with \[\ce{Cl2}\] in the presence of \[\ce{FeCl3}\].
Elimination reactions (especially β-elimination) are as common as the nucleophilic substitution reaction in case of alkyl halides. Specify the reagents used in both cases.
Which one is the correct order of nucleophilic strength (pKa) of following nucleophiles?
The number of chiral alcohol (s) with molecular formula C4H10O is ______.
The major product formed in the following reaction is:

Among the following compounds I - IV, which one forms a yellow precipitate on reacting sequentially with (i) NaOH (ii) dil. HNO3 (iii) AgNO3?
 |
 |
 |
 |
| I | II | III | IV |
In SN1 reactions, the correct order of reactivity for the following compounds:
CH3Cl, CH3CH2Cl, (CH3)2CHCl and (CH3)3CCl is ______.
Identify the product in the following reaction: 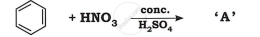
The compound that will undergo SN1 reaction with the fastest rate is:
