Advertisements
Advertisements
Question
Write the isomers of the compound having the formula C4H9Br.
Advertisements
Solution
C4H9Br is a saturated compound because its parent hydrocarbon is C4H10. Its isomers are as follows –
(i) \[\ce{\underset{1-Bromobutane}{CH3 - CH2 - CH2 - CH2 - Br}}\]
(ii) \[\begin{array}{cc}
\phantom{.......}\ce{Br}\\
\phantom{.....}|\\
\ce{\underset{2-Bromobutane}{CH3-CH2-CH-CH3}}
\end{array}\]
(iii) \[\begin{array}{cc}
\ce{CH3}\phantom{....}\\
|\phantom{.......}\\
\ce{\underset{1-Bromo-2-Methylpropane}{CH3-CH-CH2Br}}
\end{array}\]
(iv) \[\begin{array}{cc}
\ce{CH3}\\
|\phantom{...}\\
\ce{CH3 - C - Br}\phantom{.....}\\
|\phantom{...}\\
\phantom{.}\ce{\underset{2-Bromo-2-Methylpropane}{CH3}}
\end{array}\]
APPEARS IN
RELATED QUESTIONS
Which would undergo SN1 reaction faster in the following pair and why?

Write the structure of the major organic product in the following reaction:
\[\ce{CH3CH(Br)CH2CH3 + NaOH ->[water]}\]
Arrange the compounds of the following set in order of reactivity towards SN2 displacement:
1-Bromo-3-methylbutane, 2-Bromo-2-methylbutane, 2-Bromo-3-methylbutane
Arrange the compounds of the following set in order of reactivity towards SN2 displacement:
1-Bromobutane, 1-Bromo-2, 2-dimethylpropane, 1-Bromo-2-methylbutane, 1-Bromo-3-methylbutane
Identify 'A' in the following reaction -

(a) 2- Bromo-2 methylbutane
(b) 1 -Bromo-2,2-dimethylpropane
(c) 1 - Bromo - 3 -methylbutane
(d) 1 - Bromo- 2 -methylpropane
What is the action of the following on ethyl bromide
alcoholic solution of potassium hydroxide.
What is the action of the following on ethyl bromide:
moist silver oxide
In a coordination entity of the type [PtCl2(en)2]2+ which isomer will show optical isomerism?
Isopropyl chloride undergoes hydrolysis by:
The process of separation of a racemic modification into d and l-enantiomers is called ____________.
Which of the following compounds is optically active?
An organic molecule necessarily shows optical activity if it ____________.
Which of the following undergoes nucleophilic substitution exclusively by SN1 mechanism?
Which of the following statements are correct about this reaction?

(i) The given reaction follows SN2 mechanism.
(ii) (b) and (d) have opposite configuration.
(iii) (b) and (d) have same configuration.
(iv) The given reaction follows SN1 mechanism.
Which of the compounds will react faster in SN1 reaction with the –OH ion?
\[\ce{CH3-CH2-Cl}\] or \[\ce{C6H5-CH2-Cl}\]
Match the reactions given in Column I with the types of reactions given in Column II.
| Column I | Column II | |
| (i) | 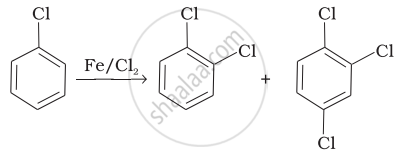 |
(a) Nucleophilic aromatic substitution |
| (ii) | \[\begin{array}{cc} \ce{CH3 - CH = CH2 + HBr -> CH3 - CH - CH3}\\ \phantom{............................}|\phantom{}\\ \phantom{.............................}\ce{Br}\phantom{} \end{array}\] |
(b) Electrophilic aromatic substitution |
| (iii) | 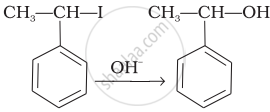 |
(c) Saytzeff elimination |
| (iv) | 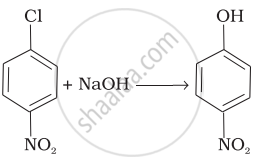 |
(d) Electrophilic addition |
| (v) | \[\begin{array}{cc} \ce{CH3 CH2 CH CH3 ->[alc.KOH] CH3 CH = CH CH3}\\ \phantom{}|\phantom{..........................}\\ \phantom{}\ce{Br}\phantom{........................} \end{array}\] |
(e) Nucleophilic substitution (SN1) |
Give the mechanism of the following reaction:
\[\ce{CH3CH2OH ->[H2SO4][413 K] CH3CH2-O-CH2CH3 + H2O}\]
Which one of the following chlorohydrocarbons readily undergoes solvolysis?
The compound that will undergo SN1 reaction with the fastest rate is:
