Advertisements
Advertisements
Question
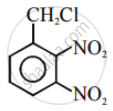
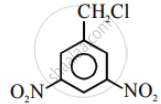
In the following pair of halogen compounds, which compound undergoes a faster SN1 reaction?

Advertisements
Solution
The activity of halogen compounds in SN1 reaction depends on the stability of the carbocation formed due to ionization. The order of stability is tertiary > secondary > primary. Hence, 2° alkyl chloride is more active than 1° alkyl chloride. Hence, 2° alkyl chloride is more active in SN1 reaction.
The halogen which reacts faster due to the greater stability of the 2° carbocation than the 1° carbocation is:

APPEARS IN
RELATED QUESTIONS
Given reasons: C–Cl bond length in chlorobenzene is shorter than C–Cl bond length in CH3–Cl.
Write the isomers of the compound having the formula C4H9Br.
What happens when methyl chloride is treated with KCN?
Which would undergo SN2 reaction faster in the following pair and why ?
CH3 – CH2 – Br and CH3 – CH2 – I
Which compound in the following pair reacts faster in SN2 reaction with OH–?
- CH3Br or CH3
- CH3Cl, (CH3)3CCl
Arrange the following organic compounds in descending order of their reactivity towards SN1 reaction.
C6H5CH2Br, C6H5CH(C6H5)Br, C6H5CH(CH3)Br, C6H5C(CH3)(C6H5)Br
Which of the following is an example of SN2 reaction?
Isopropyl chloride undergoes hydrolysis by:
The correct order of increasing the reactivity of C–X bond towards nucleophile in following compounds.

(I)

(II)
(CH3)3CCl
(III)
(CH3)2CHCl
(IV)
Ethylene chloride and ethylidene chloride are isomers. Identify the correct statements.
(i) Both the compounds form same product on treatment with alcoholic KOH.
(ii) Both the compounds form same product on treatment with aq.NaOH.
(iii) Both the compounds form same product on reduction.
(iv) Both the compounds are optically active.
Elimination reactions (especially β-elimination) are as common as the nucleophilic substitution reaction in case of alkyl halides. Specify the reagents used in both cases.
Chlorination of alkanes is an example of
CCl4 is insoluble in water because:-
Give reason for the following:
The product formed during SN1 reaction is a racemic mixture.
Optical activity of an enantiomeric mixture is +12.6° and the specific rotation of (+) isomer is +30°. The optical purity is ______ %.
The decreasing order of reactivity of the following compounds towards nucleophilic substitution (SN2) is ______.
Arrange the following compounds in increasing order of reactivity towards SN2 reaction.
2-Bromopentane, 1-Bromopentane, 2-Bromo-2-methylbutane
Complete the reaction with the main product formed:

Consider the reactions,
(i) \[\begin{array}{cc}
\phantom{}\ce{\underset{}{(CH3)2CH - CH2Br} ->[C2H5OH] \underset{}{(CH3)2CH - CH2OC2H5 + HBr}}\\
\end{array}\]
(ii) \[\begin{array}{cc}
\phantom{}\ce{\underset{}{(CH3)2CH - CH2Br} ->[C2H5O-] \underset{}{(CH3)2CH - CH2OC2H5 + Br-}}\\
\end{array}\]
The mechanisms of reactions (i) and (ii) are respectively: