Advertisements
Advertisements
Question
Give reason for the following:
The product formed during SN1 reaction is a racemic mixture.
Advertisements
Solution
During the SN1 mechanism, intermediate carbocation formed is sp2 hybridized and planar in nature. This allows the attack of nucleophiles from either side of the plane resulting in a racemic mixture.
APPEARS IN
RELATED QUESTIONS
Which would undergo SN2 reaction faster in the following pair and why ?

Arrange the compounds of the following set in order of reactivity towards SN2 displacement:
1-Bromo-3-methylbutane, 2-Bromo-2-methylbutane, 2-Bromo-3-methylbutane
C–Cl bond length in chlorobenzene is shorter than C–Cl bond length in CH3–Cl.
Given reasons: SN1 reactions are accompanied by racemization in optically active alkyl halides.
Which of the following undergoes nucleophilic substitution exclusively by SN1 mechanism?
Which of the following compound will undergo racemisation when reacts with aq. KOH?
(i)
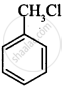
(ii)
CH3CH2CH2Cl
(iii)
\[\begin{array}{cc}
\ce{CH3}\phantom{..}\\
|\phantom{....}\\
\ce{CH3-CH-CH2Cl}
\end{array}\]
(iv)
\[\begin{array}{cc}
\phantom{..}\ce{H}\\
\phantom{..}|\\
\ce{CH3-C-Cl}\\
\phantom{..}|\\
\phantom{.....}\ce{C2H5}
\end{array}\]
A primary alkyl halide would prefer to undergo ______.
Which of the following statements are correct about the kinetics of this reaction?

(i) The rate of reaction depends on the concentration of only (b).
(ii) The rate of reaction depends on concentration of both (a) and (b).
(iii) Molecularity of reaction is one.
(iv) Molecularity of reaction is two.
Which of the compounds will react faster in SN1 reaction with the –OH ion?
\[\ce{CH3-CH2-Cl}\] or \[\ce{C6H5-CH2-Cl}\]
Consider the reactions,
(i) \[\begin{array}{cc}
\phantom{}\ce{\underset{}{(CH3)2CH - CH2Br} ->[C2H5OH] \underset{}{(CH3)2CH - CH2OC2H5 + HBr}}\\
\end{array}\]
(ii) \[\begin{array}{cc}
\phantom{}\ce{\underset{}{(CH3)2CH - CH2Br} ->[C2H5O-] \underset{}{(CH3)2CH - CH2OC2H5 + Br-}}\\
\end{array}\]
The mechanisms of reactions (i) and (ii) are respectively:
