Advertisements
Advertisements
प्रश्न
What happens when chlorobenzene is subjected to hydrolysis?
Advertisements
उत्तर
Chlorobenzene does not undergo hydrolysis under normal conditions. However, it undergoes hydrolysis when heated in an aqueous sodium hydroxide solution at 623 K and 300 atm, forming phenol. The presence of an electron-withdrawing group increases the reactivity of haloarenes.
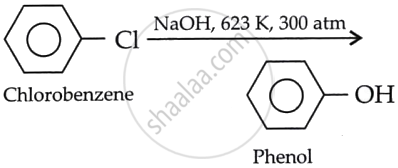
APPEARS IN
संबंधित प्रश्न
Write the major products(s) in the following:

Which would undergo SN1 reaction faster in the following pair and why?

Out of  , which is more reactive towards SN1 reaction and why?
, which is more reactive towards SN1 reaction and why?
Arrange the compounds of the following set in order of reactivity towards SN2 displacement:
2-Bromo-2-methylbutane, 1-Bromopentane, 2-Bromopentane
The stability order for carbocation is _______.
(A) 2° > 3° > 1°
(B) 3° > 2° > 1°
(C) 3° > 1° > 2°
(D) 1° > 3° > 2°
Arrange the following organic compounds in descending order of their reactivity towards SN1 reaction.
C6H5CH2Br, C6H5CH(C6H5)Br, C6H5CH(CH3)Br, C6H5C(CH3)(C6H5)Br
Halogenation of alkanes is ____________.
Which of the following undergoes nucleophilic substitution exclusively by SN1 mechanism?
Which of the following compound will undergo racemisation when reacts with aq. KOH?
(i)
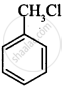
(ii)
CH3CH2CH2Cl
(iii)
\[\begin{array}{cc}
\ce{CH3}\phantom{..}\\
|\phantom{....}\\
\ce{CH3-CH-CH2Cl}
\end{array}\]
(iv)
\[\begin{array}{cc}
\phantom{..}\ce{H}\\
\phantom{..}|\\
\ce{CH3-C-Cl}\\
\phantom{..}|\\
\phantom{.....}\ce{C2H5}
\end{array}\]
The reaction of C6H5–CH=CH–CH3 with HBr produces:
Which of the following statements are correct about the kinetics of this reaction?

(i) The rate of reaction depends on the concentration of only (b).
(ii) The rate of reaction depends on concentration of both (a) and (b).
(iii) Molecularity of reaction is one.
(iv) Molecularity of reaction is two.
Which one is the correct order of nucleophilic strength (pKa) of following nucleophiles?
The number of chiral alcohol (s) with molecular formula C4H10O is ______.
Arrange the following compounds in increasing order of reactivity towards SN2 reaction.
2-Bromopentane, 1-Bromopentane, 2-Bromo-2-methylbutane
An organic compound A with the molecular formula (+) C4H9Br undergoes hydrolysis to form (+) C4H9OH. Give the structure of A and write the mechanism of the reaction.
Discuss SN2 mechanism of methyl bromide using aqueous KOH.
Which one of the following chlorohydrocarbons readily undergoes solvolysis?
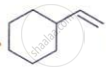 HCI, Major product ______.
HCI, Major product ______.
Which of the following is halogen exchange reaction?
