Advertisements
Advertisements
प्रश्न
Which of the following statements are correct about the kinetics of this reaction?

(i) The rate of reaction depends on the concentration of only (b).
(ii) The rate of reaction depends on concentration of both (a) and (b).
(iii) Molecularity of reaction is one.
(iv) Molecularity of reaction is two.
Advertisements
उत्तर
(i) The rate of reaction depends on the concentration of only (b).
(iii) Molecularity of reaction is one.
Explanation:
SN1 occurs in two steps. I, the polarized \[\ce{C - Cl}\] bond undergoes slow cleavage to produce a carbonation and a chloride ion. The carbocation thus formed is then attacked by nucleophile in step II to complete the substitution reaction. Step I is the slowest and reversible. It involves the \[\ce{C - Cl}\] bond breaking for which the energy is obtained through salvation of halide ion with the proton of protic solvent. Since the rate of reaction depends upon the slowest step, the rate of reaction depends only on the concentration of alkyl halide and not on the concentration of hydroxide ion. So, the rate-determining step is unimolecular.
APPEARS IN
संबंधित प्रश्न
Which would undergo SN2 reaction faster in the following pair and why ?

Write the structure of the major organic product in the following reaction:
\[\ce{CH3CH(Br)CH2CH3 + NaOH ->[water]}\]
Arrange the compounds of the following set in order of reactivity towards SN2 displacement:
1-Bromobutane, 1-Bromo-2, 2-dimethylpropane, 1-Bromo-2-methylbutane, 1-Bromo-3-methylbutane
What is the action of the following on ethyl bromide?
moist silver oxide
Which compound in the following pair reacts faster in SN2 reaction with OH–?
- CH3Br or CH3
- CH3Cl, (CH3)3CCl
Halogenation of alkanes is ____________.
Most reactive halide towards SN1 reaction is ____________.
Optically active isomers but not mirror images are called ____________.
Isopropyl chloride undergoes hydrolysis by:
Which of the following is a chiral compound?
Identify X and Y in the following sequence:
\[\ce{C2H5Br ->[X] Product ->[Y] C3H7NH2}\]
A primary alkyl halide would prefer to undergo ______.
Which of the following statements are correct about this reaction?

(i) The given reaction follows SN2 mechanism.
(ii) (b) and (d) have opposite configuration.
(iii) (b) and (d) have same configuration.
(iv) The given reaction follows SN1 mechanism.
Elimination reactions (especially β-elimination) are as common as the nucleophilic substitution reaction in case of alkyl halides. Specify the reagents used in both cases.
Aryl halides are extremely less reactive towards nucleophilic substitution. Predict and explain the order of reactivity of the following compounds towards nucleophilic substitution:
| (I) |  |
| (II) |  |
| (III) |  |
Chlorination of alkanes is an example of
The number of chiral alcohol (s) with molecular formula C4H10O is ______.
In SN1 reactions, the correct order of reactivity for the following compounds:
CH3Cl, CH3CH2Cl, (CH3)2CHCl and (CH3)3CCl is ______.
Acetic anhydride from acetic acid
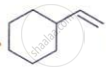 HCI, Major product ______.
HCI, Major product ______.
