Advertisements
Advertisements
प्रश्न
Assertion: KCN reacts with methyl chloride to give methyl isocyanide.
Reason: CN– is an ambident nucleophile.
पर्याय
Assertion and reason both are correct and reason is correct explanation of assertion.
Assertion and reason both are wrong statements.
Assertion is correct but reason is wrong statement.
Assertion is wrong but reason is correct statement.
Assertion and reason both are correct statements but reason is not correct explanation of assertion.
Advertisements
उत्तर
Assertion is wrong but reason is correct statement.
Explanation:
\[\ce{\underset{Alkyl cyanide}{R - Cl + KCN –> R - CN + KCl}}\]
Isocyanide is not obtained in this reaction. CN– is an ambident nucleophile.
APPEARS IN
संबंधित प्रश्न
Which would undergo SN2 reaction faster in the following pair and why ?

Arrange the compounds of the following set in order of reactivity towards SN2 displacement:
1-Bromo-3-methylbutane, 2-Bromo-2-methylbutane, 2-Bromo-3-methylbutane
Out of C6H5CH2Cl and C6H5CHClC6H5, which is more easily hydrolysed by aqueous KOH.
What happens when chlorobenzene is subjected to hydrolysis?
What happens when ethyl chloride is treated with aqueous KOH?
What is the action of the following on ethyl bromide:
moist silver oxide
In the reaction, \[\ce{R - X + NaOR' -> ROR’ + X}\] ( – ve ion). The main product formed is:
Which of the following is optically inactive?
Which one of the following halogen compounds is difficult to be hydrolysed by SN1 mechanism?
Which of the following compounds is optically active?
Which of the following is a chiral compound?
The increasing order of nucleophilicity would be:
Which of the following compound will undergo racemisation when reacts with aq. KOH?
(i)
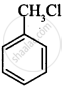
(ii)
CH3CH2CH2Cl
(iii)
\[\begin{array}{cc}
\ce{CH3}\phantom{..}\\
|\phantom{....}\\
\ce{CH3-CH-CH2Cl}
\end{array}\]
(iv)
\[\begin{array}{cc}
\phantom{..}\ce{H}\\
\phantom{..}|\\
\ce{CH3-C-Cl}\\
\phantom{..}|\\
\phantom{.....}\ce{C2H5}
\end{array}\]
The increasing order of reactivity towards SN1 mechanism is:
(I) \[\begin{array}{cc}
\ce{CH3-CH-CH2-CH3}\\
|\phantom{........}\\
\ce{CH3}\phantom{.....}
\end{array}\]
(II) CH3CH2CH2Cl
(III) P–CH3O–C6H4–CH2Cl
Which reagent will you use for the following reaction?
\[\ce{CH3CH2CH2CH3 -> CH3CH2CH2CH2Cl + CH3CH2CHClCH3}\]
Elimination reactions (especially β-elimination) are as common as the nucleophilic substitution reaction in case of alkyl halides. Specify the reagents used in both cases.
Which of the following is the definition of chirality?
In which reaction mechanism carbocation is formed?
Which one of the following chlorohydrocarbons readily undergoes solvolysis?
Assertion (A):  undergoes SN2 reactions faster than
undergoes SN2 reactions faster than  .
.
Reason (R): Iodine is a better leaving group because of its large size.
In the light of the above statements, choose the correct answer from the options given below:
