Advertisements
Advertisements
प्रश्न
Elimination reactions (especially β-elimination) are as common as the nucleophilic substitution reaction in case of alkyl halides. Specify the reagents used in both cases.
Advertisements
उत्तर
Alkyl halides undergo nucleophilic substitution as well as elimination (Beta-elimination) reaction. However, by proper choice of reagents and reaction conditions, a particular product can be obtained. Usually strong and bulky bases and high temperature favour elimination reactions while weaker and smaller bases and lower temperature favour substitution reactions. For example, ethyl bromide on heating with alcoholic KOH (which contain stronger base, \[\ce{C2H5O}\] ion) at about 473-523 K undergoes elimination to give ethene. But with aqueous \[\ce{KOH}\] at about 373 K, it gives ethanol.
\[\ce{CH3CH2Br ->[alc.KOH][473-523 K] CH2 = CH2 (Elimination)}\]
\[\ce{CH3CH2Br ->[aq.KOH][373 K] CH3CH2OH (Substitution)}\]
Nucleophilic substitution: Reagents used nucleophilies like \[\ce{- \overset{-}{O}H, NH3, \overset{-}{C} ≡ N;, AgCN:, O = N - O, \overset{-}{O}R}\]' etc. also, alc. \[\ce{KOH}\] at lower temperature (373 K) undergoes substitution reaction.
APPEARS IN
संबंधित प्रश्न
Discuss the mechanism of alkaline hydrolysis of bromomethane.
Write the major products(s) in the following:

Write the mechanism of the following reaction:
\[\ce{{n}BuBr + KCN ->[EtOH-H2O] {n}BuCN}\]
The stability order for carbocation is _______.
(A) 2° > 3° > 1°
(B) 3° > 2° > 1°
(C) 3° > 1° > 2°
(D) 1° > 3° > 2°
Which would undergo SN2 reaction faster in the following pair and why ?
CH3 – CH2 – Br and CH3 – CH2 – I
What is the action of the following on ethyl bromide?
moist silver oxide
Answer the following question.
Write one stereochemical difference between SN1 and SN2 reactions.
Arrange the following organic compounds in descending order of their reactivity towards SN1 reaction.
C6H5CH2Br, C6H5CH(C6H5)Br, C6H5CH(CH3)Br, C6H5C(CH3)(C6H5)Br
The order of reactivities of the following alkyl halides for an SN2 reaction is:
Optically active isomers but not mirror images are called ____________.
Racemic compound has ____________.
Which of the following is a chiral compound?
Ethylene chloride and ethylidene chloride are isomers. Identify the correct statements.
(i) Both the compounds form same product on treatment with alcoholic KOH.
(ii) Both the compounds form same product on treatment with aq.NaOH.
(iii) Both the compounds form same product on reduction.
(iv) Both the compounds are optically active.
Which of the compounds will react faster in SN1 reaction with the –OH ion?
\[\ce{CH3-CH2-Cl}\] or \[\ce{C6H5-CH2-Cl}\]
Optical activity of an enantiomeric mixture is +12.6° and the specific rotation of (+) isomer is +30°. The optical purity is ______ %.
Racemisation occurs in ______.
Assertion (A) : Nucleophilic substitution of iodoethane is easier than chloroethane.
Reason (R) : Bond enthalpy of C-I bond is less than that of C-Cl bond.
Identify the product in the following reaction: 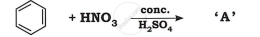
Which alkyl halide from the following pair would you expect to react more rapidly by an SN2 mechanism? Explain your answer.
\[\begin{array}{cc}\ce{CH3CH2CHCH3}\\
\phantom{.....}|\\
\phantom{......}\ce{Br}\\
\end{array}\] or \[\begin{array}{cc}\phantom{.......}\ce{CH3}\\
\phantom{...}|\\
\ce{H3C - C - Br}\\
\phantom{...}|\\
\phantom{.......}\ce{CH3}\\
\end{array}\]
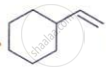 HCI, Major product ______.
HCI, Major product ______.
