Advertisements
Advertisements
प्रश्न
Elimination reactions (especially β-elimination) are as common as the nucleophilic substitution reaction in case of alkyl halides. Specify the reagents used in both cases.
Advertisements
उत्तर
Alkyl halides undergo nucleophilic substitution as well as elimination (Beta-elimination) reaction. However, by proper choice of reagents and reaction conditions, a particular product can be obtained. Usually strong and bulky bases and high temperature favour elimination reactions while weaker and smaller bases and lower temperature favour substitution reactions. For example, ethyl bromide on heating with alcoholic KOH (which contain stronger base, \[\ce{C2H5O}\] ion) at about 473-523 K undergoes elimination to give ethene. But with aqueous \[\ce{KOH}\] at about 373 K, it gives ethanol.
\[\ce{CH3CH2Br ->[alc.KOH][473-523 K] CH2 = CH2 (Elimination)}\]
\[\ce{CH3CH2Br ->[aq.KOH][373 K] CH3CH2OH (Substitution)}\]
Nucleophilic substitution: Reagents used nucleophilies like \[\ce{- \overset{-}{O}H, NH3, \overset{-}{C} ≡ N;, AgCN:, O = N - O, \overset{-}{O}R}\]' etc. also, alc. \[\ce{KOH}\] at lower temperature (373 K) undergoes substitution reaction.
APPEARS IN
संबंधित प्रश्न
Write the structure of the major product in each of the following reaction :

Which compound in the following pair will react faster in SN2 reaction with OH−?
(CH3)3CCl or CH3Cl
Write the mechanism of the following reaction:
\[\ce{{n}BuBr + KCN ->[EtOH-H2O] {n}BuCN}\]
Arrange the compounds of the following set in order of reactivity towards SN2 displacement:
1-Bromobutane, 1-Bromo-2, 2-dimethylpropane, 1-Bromo-2-methylbutane, 1-Bromo-3-methylbutane
What happens when ethyl chloride is treated with aqueous KOH?
Answer the following question.
Write one stereochemical difference between SN1 and SN2 reactions.
Which of the following is a primary halide?
Which among MeX, RCH2X, R2CHX and R3CX is most reactive towards SN2 reaction?
Which of the following compounds is optically active?
2-Bromopentane is heated with potassium ethoxide in ethanol. The major product obtained is ____________.
Which of the following is a chiral compound?
The correct order of increasing the reactivity of C–X bond towards nucleophile in following compounds.

(I)

(II)
(CH3)3CCl
(III)
(CH3)2CHCl
(IV)
Which of the following compound will undergo racemisation when reacts with aq. KOH?
(i)
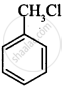
(ii)
CH3CH2CH2Cl
(iii)
\[\begin{array}{cc}
\ce{CH3}\phantom{..}\\
|\phantom{....}\\
\ce{CH3-CH-CH2Cl}
\end{array}\]
(iv)
\[\begin{array}{cc}
\phantom{..}\ce{H}\\
\phantom{..}|\\
\ce{CH3-C-Cl}\\
\phantom{..}|\\
\phantom{.....}\ce{C2H5}
\end{array}\]
Which of the following alkyl halides will undergo SN1 reaction most readily?
Which reagent will you use for the following reaction?
\[\ce{CH3CH2CH2CH3 -> CH3CH2CH2CH2Cl + CH3CH2CHClCH3}\]
The number of chiral alcohol (s) with molecular formula C4H10O is ______.
Among the following compounds I - IV, which one forms a yellow precipitate on reacting sequentially with (i) NaOH (ii) dil. HNO3 (iii) AgNO3?
 |
 |
 |
 |
| I | II | III | IV |
Complete the reaction with the main product formed:

The following questions are case-based questions. Read the passage carefully and answer the questions that follow:
|
Nucleophilic Substitution: Influences of solvent polarity: The reaction rate (SN2) of 2-bromopropane and NaOH in ethanol containing 40% water is twice slower than in absolute ethanol. Hence the level of solvent polarity has an influence on both SN1 and SN2 reactions but with different results. Generally speaking, a weak polar solvent is favourable for SN2 reaction, while a strong polar solvent is favourable for SN1. Generally speaking, the substitution reaction of tertiary haloalkane is based on SN1 mechanism in solvents with a strong polarity (for example ethanol containing water). |
Answer the following questions:
(a) Why racemisation occurs in SN1? (1)
(b) Why is ethanol less polar than water? (1)
(c) Which one of, the following in each pair is more reactive towards SN2 reaction? (2)
(i) CH3 – CH2 – I or CH3CH2 – Cl
(ii)
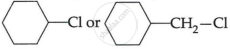
OR
(c) Arrange the following in the increasing order of their reactivity towards SN1 reactions: (2)
(i) 2-Bromo-2-methylbutane, 1-Bromo-pentane, 2-Bromo-pentane
(ii) 1-Bromo-3-methylbutane, 2-Bromo-2-methylbutane, 2-Bromo-3- methylbutane
Which of the following is halogen exchange reaction?
