Advertisements
Advertisements
प्रश्न
What is the action of the following on ethyl bromide
alcoholic solution of potassium hydroxide.
Advertisements
उत्तर
\[\ce{\underset{\text{ethylbromide}}{C2H5Br} + alc KOH ->\underset{\text{ethene}}{CH2} = CH2 + KBr + H2O}\]
APPEARS IN
संबंधित प्रश्न
Write the structure of an isomer of compound C4H9Br which is most reactive towards SN1 reaction
Write the isomers of the compound having the formula C4H9Br.
What are ambident nucleophiles? Explain with an example.
Arrange the compounds of the following set in order of reactivity towards SN2 displacement:
2-Bromo-2-methylbutane, 1-Bromopentane, 2-Bromopentane
Arrange the compounds of the following set in order of reactivity towards SN2 displacement:
1-Bromo-3-methylbutane, 2-Bromo-2-methylbutane, 2-Bromo-3-methylbutane
What happens when methyl chloride is treated with KCN?
Which would undergo SN2 reaction faster in the following pair and why ?
CH3 – CH2 – Br and CH3 – CH2 – I
What is the action of the following on ethyl bromide?
silver acetate
Answer the following question.
Write one stereochemical difference between SN1 and SN2 reactions.
Which of the following is optically inactive?
Which one is most reactive towards SN1 reaction?
Isopropyl chloride undergoes hydrolysis by:
The process of separation of a racemic modification into d and l-enantiomers is called ____________.
Identify the end product (C) in the following sequence:
\[\ce{C2H5OH ->[SOCl2][Pyridine] A ->[KCN {(alc.)}] B ->[2H2O/H^+] C}\]
Which of the following undergoes nucleophilic substitution exclusively by SN1 mechanism?
The increasing order of nucleophilicity would be:
The correct order of increasing the reactivity of C–X bond towards nucleophile in following compounds.

(I)

(II)
(CH3)3CCl
(III)
(CH3)2CHCl
(IV)
Which of the following compounds will give a racemic mixture on nucleophilic substitution by OH ion?
1-Bromoethane, 1-Bromopropane, 1-Bromobutane, Bromobenzene

Which of the statements are correct about above reaction?
(i) (a) and (e) both are nucleophiles.
(ii) In (c) carbon atom is sp3 hybridised.
(iii) In (c) carbon atom is sp2 hybridised.
(iv) (a) and (e) both are electrophiles.
Write the structures and names of the compounds formed when compound ‘A’ with molecular formula, \[\ce{C7H8}\] is treated with \[\ce{Cl2}\] in the presence of \[\ce{FeCl3}\].
How do polar solvents help in the first step in SN1 mechanism?
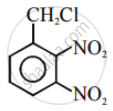
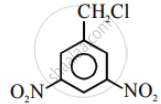
Aryl halides are extremely less reactive towards nucleophilic substitution. Predict and explain the order of reactivity of the following compounds towards nucleophilic substitution:
| (I) |  |
| (II) |  |
| (III) |  |
Cyanide ion acts as an ambident nucleophile. From which end it acts as a stronger nucleophile in aqueous medium? Give reason for your answer.
Which one of the following compounds is more reactive towards SN1 reaction?
The decreasing order of reactivity of the following compounds towards nucleophilic substitution (SN2) is ______.
Racemisation occurs in ______.
Inversion of configuration occurs in ______.
Complete the reaction with the main product formed:

Convert bromoethane to propanamine.