Advertisements
Advertisements
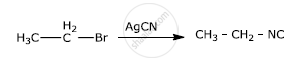
प्रश्न
Write the major products(s) in the following:

Advertisements
उत्तर
APPEARS IN
संबंधित प्रश्न
Write the structure of the major product in each of the following reaction :

Which compound in the following pair will react faster in SN2 reaction with OH−?
(CH3)3CCl or CH3Cl
Which of the following is an optically active compound?
2-Bromopentane is heated with potassium ethoxide in ethanol. The major product obtained is ____________.
Ethylene chloride and ethylidene chloride are isomers. Identify the correct statements.
(i) Both the compounds form same product on treatment with alcoholic KOH.
(ii) Both the compounds form same product on treatment with aq.NaOH.
(iii) Both the compounds form same product on reduction.
(iv) Both the compounds are optically active.
Write the structures and names of the compounds formed when compound ‘A’ with molecular formula, \[\ce{C7H8}\] is treated with \[\ce{Cl2}\] in the presence of \[\ce{FeCl3}\].
The number of chiral alcohol (s) with molecular formula C4H10O is ______.
Identify the product in the following reaction: 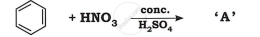
Consider the reactions,
(i) \[\begin{array}{cc}
\phantom{}\ce{\underset{}{(CH3)2CH - CH2Br} ->[C2H5OH] \underset{}{(CH3)2CH - CH2OC2H5 + HBr}}\\
\end{array}\]
(ii) \[\begin{array}{cc}
\phantom{}\ce{\underset{}{(CH3)2CH - CH2Br} ->[C2H5O-] \underset{}{(CH3)2CH - CH2OC2H5 + Br-}}\\
\end{array}\]
The mechanisms of reactions (i) and (ii) are respectively:
The correct order of increasing reactivity of
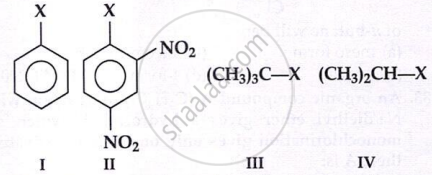
C-X bond towards nucleophile in the following compounds is:
