Advertisements
Advertisements
प्रश्न
The correct order of increasing reactivity of
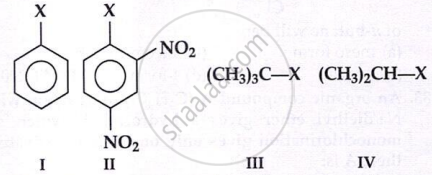
C-X bond towards nucleophile in the following compounds is:
पर्याय
I < II < IV < III
II < III < I < IV
IV < III > I > II
III > II < I < IV
Advertisements
उत्तर
I < II < IV < III
Explanation:
The order of reactivity is dependent on the stability of the intermediate carbocation formed by cleavage of C − X bond. The 3° carbocation formed in III will be more stable than its 2° homologue (IV) which in turn will be more stable than arenium ion (I). Moreover, the aryl halide has a double bond character in the C − X bond which makes the cleavage more difficult.
On other hand II will be more reactive than I due to presence of electron withdrawing −NO2 group. C − X bond becomes weak and undergoes nucleophilic substitution reaction.
∴ Order: I < II < IV < III.
