Advertisements
Advertisements
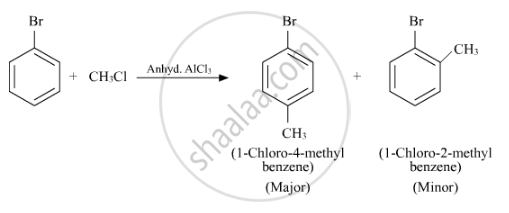
प्रश्न
Write the structure of the major product in each of the following reaction :

Advertisements
उत्तर
संबंधित प्रश्न
How do you convert the following:
Ethanol to propanenitrile
SN1 reactions are accompanied by racemization in optically active alkyl halides.
Given reasons: SN1 reactions are accompanied by racemization in optically active alkyl halides.
Which would undergo SN2 reaction faster in the following pair and why ?
CH3 – CH2 – Br and CH3 – CH2 – I
The order of reactivity of the given haloalkanes towards nucleophile is:
2-Bromopentane is heated with potassium ethoxide in ethanol. The major product obtained is ____________.
Among the following compounds I - IV, which one forms a yellow precipitate on reacting sequentially with (i) NaOH (ii) dil. HNO3 (iii) AgNO3?
 |
 |
 |
 |
| I | II | III | IV |
Optical activity of an enantiomeric mixture is +12.6° and the specific rotation of (+) isomer is +30°. The optical purity is ______ %.
Assertion (A) : Nucleophilic substitution of iodoethane is easier than chloroethane.
Reason (R) : Bond enthalpy of C-I bond is less than that of C-Cl bond.
The correct order of increasing reactivity of
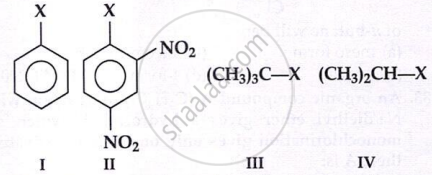
C-X bond towards nucleophile in the following compounds is:
