Advertisements
Advertisements
प्रश्न
Write the structures and names of the compounds formed when compound ‘A’ with molecular formula, \[\ce{C7H8}\] is treated with \[\ce{Cl2}\] in the presence of \[\ce{FeCl3}\].
Advertisements
उत्तर
The compound with molecular formula \[\ce{C7H8}\] is toluene, \[\ce{C6H5CH3}\]. Since \[\ce{^-CH3}\] group is o-, p-directing, therefore, chlorination of toluene gives o-chlorotoluene and p-chlorotoluene, in which the p-isomer predominates.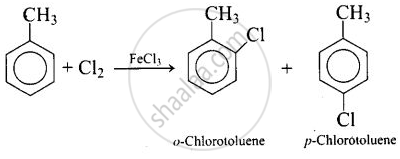
APPEARS IN
संबंधित प्रश्न
In the following pair of halogen compounds, which compound undergoes a faster SN1 reaction?

In the following pair of halogen compounds, which compound undergoes a faster SN1 reaction?

C–Cl bond length in chlorobenzene is shorter than C–Cl bond length in CH3–Cl.
Identify 'A' in the following reaction -

(a) 2- Bromo-2 methylbutane
(b) 1 -Bromo-2,2-dimethylpropane
(c) 1 - Bromo - 3 -methylbutane
(d) 1 - Bromo- 2 -methylpropane
What is the action of the following on ethyl bromide?
silver acetate
In the reaction, \[\ce{R - X + NaOR' -> ROR’ + X}\] ( – ve ion). The main product formed is:
Which of the following is a primary halide?
Which one is most reactive towards SN1 reaction?
Which one of the following halogen compounds is difficult to be hydrolysed by SN1 mechanism?
The order of reactivity of the given haloalkanes towards nucleophile is:
Most reactive halide towards SN1 reaction is ____________.
The process of separation of a racemic modification into d and l-enantiomers is called ____________.
Identify X and Y in the following sequence:
\[\ce{C2H5Br ->[X] Product ->[Y] C3H7NH2}\]
Which of the following undergoes nucleophilic substitution exclusively by SN1 mechanism?
Which of the following statements are correct about this reaction?

(i) The given reaction follows SN2 mechanism.
(ii) (b) and (d) have opposite configuration.
(iii) (b) and (d) have same configuration.
(iv) The given reaction follows SN1 mechanism.
Which of the following is the definition of chirality?
Which alkyl halide from the following pair would you expect to react more rapidly by an SN2 mechanism? Explain your answer.
\[\begin{array}{cc}\ce{CH3CH2CHCH3}\\
\phantom{.....}|\\
\phantom{......}\ce{Br}\\
\end{array}\] or \[\begin{array}{cc}\phantom{.......}\ce{CH3}\\
\phantom{...}|\\
\ce{H3C - C - Br}\\
\phantom{...}|\\
\phantom{.......}\ce{CH3}\\
\end{array}\]
Which of the following is halogen exchange reaction?
Which of the following reactions is an example of nucleophilic substitution reaction?
