Advertisements
Advertisements
प्रश्न
Write the structures and names of the compounds formed when compound ‘A’ with molecular formula, \[\ce{C7H8}\] is treated with \[\ce{Cl2}\] in the presence of \[\ce{FeCl3}\].
Advertisements
उत्तर
The compound with molecular formula \[\ce{C7H8}\] is toluene, \[\ce{C6H5CH3}\]. Since \[\ce{^-CH3}\] group is o-, p-directing, therefore, chlorination of toluene gives o-chlorotoluene and p-chlorotoluene, in which the p-isomer predominates.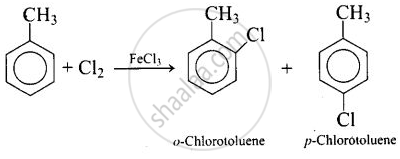
APPEARS IN
संबंधित प्रश्न
Which would undergo SN1 reaction faster in the following pair and why?

Which alkyl halide from the following pair would you expect to react more rapidly by an SN2 mechanism? Explain your answer.
CH3CH2CH2CH2Br or \[\begin{array}{cc}
\ce{CH3CH2CHCH3}\\
\phantom{...}|\\
\phantom{....}\ce{Br}\
\end{array}\]
What happens when ethyl chloride is treated with aqueous KOH?
What happens when methyl chloride is treated with KCN?
The stability order for carbocation is _______.
(A) 2° > 3° > 1°
(B) 3° > 2° > 1°
(C) 3° > 1° > 2°
(D) 1° > 3° > 2°
What is the action of the following on ethyl bromide
alcoholic solution of potassium hydroxide.
What is the action of the following on ethyl bromide?
moist silver oxide
Arrange the following organic compounds in descending order of their reactivity towards SN1 reaction.
C6H5CH2Br, C6H5CH(C6H5)Br, C6H5CH(CH3)Br, C6H5C(CH3)(C6H5)Br
Which of the following is optically inactive?
Halogenation of alkanes is ____________.
The order of reactivities of the following alkyl halides for an SN2 reaction is:
An important chemical method to resolve a racemic mixture makes use of the formation of ______.
Racemic compound has ____________.

Which of the statements are correct about above reaction?
(i) (a) and (e) both are nucleophiles.
(ii) In (c) carbon atom is sp3 hybridised.
(iii) In (c) carbon atom is sp2 hybridised.
(iv) (a) and (e) both are electrophiles.
Elimination reactions (especially β-elimination) are as common as the nucleophilic substitution reaction in case of alkyl halides. Specify the reagents used in both cases.
Match the reactions given in Column I with the types of reactions given in Column II.
| Column I | Column II | |
| (i) | 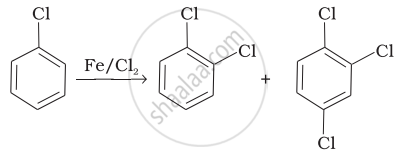 |
(a) Nucleophilic aromatic substitution |
| (ii) | \[\begin{array}{cc} \ce{CH3 - CH = CH2 + HBr -> CH3 - CH - CH3}\\ \phantom{............................}|\phantom{}\\ \phantom{.............................}\ce{Br}\phantom{} \end{array}\] |
(b) Electrophilic aromatic substitution |
| (iii) | 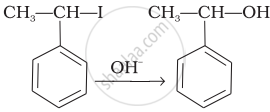 |
(c) Saytzeff elimination |
| (iv) | 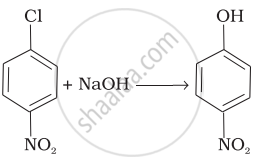 |
(d) Electrophilic addition |
| (v) | \[\begin{array}{cc} \ce{CH3 CH2 CH CH3 ->[alc.KOH] CH3 CH = CH CH3}\\ \phantom{}|\phantom{..........................}\\ \phantom{}\ce{Br}\phantom{........................} \end{array}\] |
(e) Nucleophilic substitution (SN1) |
Inversion of configuration occurs in ______.
Acetic anhydride from acetic acid
Which one of the following chlorohydrocarbons readily undergoes solvolysis?
