Advertisements
Advertisements
प्रश्न
Which alkyl halide from the following pair would you expect to react more rapidly by an SN2 mechanism? Explain your answer.
CH3CH2CH2CH2Br or \[\begin{array}{cc}
\ce{CH3CH2CHCH3}\\
\phantom{...}|\\
\phantom{....}\ce{Br}\
\end{array}\]
Advertisements
उत्तर
The SN2 process involves a transition state with both an incoming nucleophile and a leaving group surrounding the carbon atom. Five atoms are simultaneously bonded together. A transition state requires minimal steric hindrance. Hence, 1° alkyl halides are the most reactive to SN2, followed by 2° and 3°.
1° RX > 2° RX > 3° RX
Based on the above order, CH3CH2CH2CH2Br is more reactive.
APPEARS IN
संबंधित प्रश्न
Write the structure of an isomer of compound C4H9Br which is most reactive towards SN1 reaction
Write the main products when methyl chloride is treated with AgCN.
In the following pair of halogen compounds, which compound undergoes a faster SN1 reaction?

How the following conversion can be carried out?
Ethyl chloride to propanoic acid.
Which of the following is an example of SN2 reaction?
Which of the following is a primary halide?
Optically active isomers but not mirror images are called ____________.
Which among MeX, RCH2X, R2CHX and R3CX is most reactive towards SN2 reaction?
Which of the following is the correct order of decreasing SN2 reactivity?
SN1 reaction of alkyl halides lead to ___________.
A primary alkyl halide would prefer to undergo ______.

Which of the statements are correct about above reaction?
(i) (a) and (e) both are nucleophiles.
(ii) In (c) carbon atom is sp3 hybridised.
(iii) In (c) carbon atom is sp2 hybridised.
(iv) (a) and (e) both are electrophiles.
Cyanide ion acts as an ambident nucleophile. From which end it acts as a stronger nucleophile in aqueous medium? Give reason for your answer.
Chlorination of alkanes is an example of
In SN1 reactions, the correct order of reactivity for the following compounds:
CH3Cl, CH3CH2Cl, (CH3)2CHCl and (CH3)3CCl is ______.
Identify the product in the following reaction: 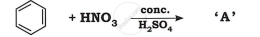
Which alkyl halide from the following pair would you expect to react more rapidly by an SN2 mechanism? Explain your answer.
\[\begin{array}{cc}\ce{CH3CH2CHCH3}\\\phantom{...}|\\\phantom{....}\ce{Br}\end{array}\] or \[\begin{array}{cc}\phantom{.....}\ce{CH3}\\\phantom{..}|\\\ce{H3C - C - Br}\\\phantom{..}|\\\phantom{....}\ce{CH3}\end{array}\]
Which alkyl halide from the following pair would you expect to react more rapidly by an SN2 mechanism? Explain your answer.
\[\begin{array}{cc}\ce{CH3CHCH2CH2Br}\\|\phantom{.........}\\\ce{CH3}\phantom{......}\end{array}\] or \[\begin{array}{cc}\ce{CH3CH2CHCH2Br}\\\phantom{}|\\\phantom{...}\ce{CH3}\end{array}\]
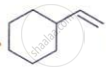 HCI, Major product ______.
HCI, Major product ______.
