Advertisements
Advertisements
प्रश्न
Arrange the set of compounds in order of increasing boiling points.
1-Chloropropane, Isopropyl chloride, 1-Chlorobutane.
Advertisements
उत्तर
Molecules with the same mass have different boiling points based on their size. Branched compounds have a lower boiling point than straight-chain compounds due to their smaller surface area and compact structure. The order of boiling points is:
Isopropyl chloride < 1-Chloropropane < 1-Chlorobutane
APPEARS IN
संबंधित प्रश्न
Arrange the set of compounds in order of increasing boiling points.
Bromomethane, Bromoform, Chloromethane, Dibromomethane.
Explain why alkyl halides, though polar, are immiscible with water?
p-Dichlorobenzene has higher m.p. and lower solubility than those of o- and m-isomers. Discuss.
Define racemic mixture.
For the same alkyl group, an alkyl bromide has a higher boiling point than alkyl fluoride because:
Which of the following compounds has the highest boiling point?
How many structural isomers are possible for a compound with the molecular formula C3H7Cl?
Which of the following halide is 2°?
The decreasing order of boiling points of alkyl halides is:
Arrange the following compounds in increasing order of their boiling points.
(a) \[\begin{array}{cc}
\ce{CH3}\phantom{..................}\\
\backslash\phantom{...........}\\
\ce{CH - CH - CH2Br}\\
/\phantom{.............}\\
\ce{CH3}\phantom{..................}
\end{array}\]
(b) \[\ce{CH3CH2CH2CH2Br}\]
(c) \[\begin{array}{cc}
\phantom{...}\ce{CH3}\\
\phantom{}|\\
\ce{H3C - C - CH3}\\
\phantom{}|\\
\phantom{..}\ce{Br}
\end{array}\]
Reaction of \[\ce{C6H5CH2Br}\] with aqueous sodium hydroxide follows ______.
Which is the correct increasing order of boiling points of the following compounds?
1-Iodobutane, 1-Bromobutane, 1-Chlorobutane, Butane
Which is the correct increasing order of boiling points of the following compounds?
1-Bromoethane, 1-Bromopropane, 1-Bromobutane, Bromobenzene
Out of o-and p-dibromobenzene which one has higher melting point and why?
Which of the following compounds will have the highest melting point and why?
| (I) |  |
|
(II) |
 |
| (III) | 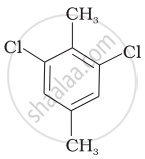 |
Why is the boiling point of o-dichlorobenzene higher than p-dichlorobenzene, but the melting point of para-isomer is higher than ortho-isomer?
Arrange the isomeric dichlorobenzene in the increasing order of their boiling point and melting points.
Name the following halides according to the IUPAC system and classify them as alkyl, allyl, benzyl (primary, secondary, tertiary), vinyl or aryl halide:
\[\ce{CH3 C(C2H5)2CH2Br}\]
Write the structure of the following organic halogen compound.
4-tert-Butyl-3-iodoheptane
