Advertisements
Advertisements
प्रश्न
Arrange the set of compounds in order of increasing boiling points.
Bromomethane, Bromoform, Chloromethane, Dibromomethane.
Advertisements
उत्तर
The strength of the intermolecular interactions in organic molecules determines their boiling points. These forces consist of van der Waals forces and dipole-dipole interactions and depend on the molecular mass and surface area of the molecules. Compounds with higher molecular mass have higher boiling points. Thus, the correct order is:
Chloromethane < Bromomethane < Dibromomethane < Bromoform
संबंधित प्रश्न
Explain why alkyl halides, though polar, are immiscible with water?
Define racemic mixture.
For the same alkyl group, an alkyl bromide has a higher boiling point than alkyl fluoride because:
Which of the following compounds has the highest boiling point?
The decreasing order of boiling points of alkyl halides is:
Which of the following is liquid at room temperature (b.p. is shown against it)?
Which of the following possesses the highest melting point?
Mg reacts with RBr best in ____________.
p-dichlorobenzene has a higher melting point than its o- and m-isomers because ____________.
Arrange the following compounds in the increasing order of their densities.
(a)

(b)

(c)
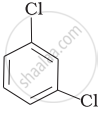
(d)

Reaction of \[\ce{C6H5CH2Br}\] with aqueous sodium hydroxide follows ______.
Out of o-and p-dibromobenzene which one has higher melting point and why?
Arrange the following compounds in increasing order of their boiling points:
CH3CH2OH, CH3−CHO, CH3−COOH
Why alkyl halides though polar are immiscible with water?
Write the structure of the following organic halogen compound.
1,4-Dibromobut-2-ene
Write the structure of the following organic halogen compound:
4-tert-Butyl-3-iodoheptane
Name the following halides according to the IUPAC system and classify them as alkyl, allyl, benzyl (primary, secondary, tertiary), vinyl or aryl halide:
\[\ce{CH3 C(C2H5)2CH2Br}\]
Write the structure of the following organic halogen compound.
4-tert-Butyl-3-iodoheptane
