Advertisements
Advertisements
प्रश्न
Compound ‘A’ with molecular formula \[\ce{C4H9Br}\] is treated with aq. \[\ce{KOH}\] solution. The rate of this reaction depends upon the concentration of the compound ‘A’ only. When another optically active isomer ‘B’ of this compound was treated with aq. \[\ce{KOH}\] solution, the rate of reaction was found to be dependent on concentration of compound and \[\ce{KOH}\] both.
(i) Write down the structural formula of both compounds ‘A’ and ‘B’.
(ii) Out of these two compounds, which one will be converted to the product with inverted configuration.
Advertisements
उत्तर
(i) The molecular formulae of isomers of \[\ce{C4H9Br}\] are \[\ce{CH3}\]
\[\begin{array}{cc}
\ce{CH3}\phantom{......}\\
|\phantom{.........}\\
\ce{CH3 - C - Br}\phantom{...........}\\
|\phantom{.........}\\
\ce{\underset{2 - Bromo-2-methyl propane (A)}{CH3}}\phantom{......}
\end{array}\]
\[\begin{array}{cc}
\ce{CH3 - CH2 - CH - CH3}\\
\phantom{.....}|\phantom{}\\
\phantom{.......}\ce{\underset{2-Bromobutance (B)}{Br}}\phantom{}
\end{array}\]
Since the rate of reaction of compound ‘A’ \[\ce{(C4H9Br)}\] with aqueous \[\ce{KOH}\] depends upon the concentration of compound ‘A’ only, therefore, the reaction occurs by SN1 mechanism and compound ‘A’ is tertiary bromide i.e., 2-Bromo-2-methylpropane.
\[\ce{(CH3)3CBr + KOH(aq) –> (CH3)3COH + KBr}\]
Rate = \[\ce{A:[(CH3)3CBr]}\]
(ii) Since compound ‘B’ is optically active and is an isomer of compound ‘A’ \[\ce{(C4H9Br)}\], therefore, compound ‘B’ must be 2-Bromobutane. Since the rate of reaction of compound ‘B’ with aqueous \[\ce{KOH}\] depends upon the concentration of compound ‘B’ and \[\ce{KOH}\], therefore, the reaction occurs by SN2 mechanism and product of hydrolysis will have inverted configuration.
\[\begin{array}{cc}
\ce{CH3CH2CHCH3 + KOH -> CH3CH2CHCH3 + KBr}\\
\phantom{..}|\phantom{...............................}|\phantom{...}\\
\phantom{..}\ce{Br}\phantom{.............................}\ce{OH}\phantom{.}
\end{array}\]
Rate = 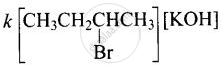
APPEARS IN
संबंधित प्रश्न
Out of  , which is more reactive towards SN1 reaction and why?
, which is more reactive towards SN1 reaction and why?
What are ambident nucleophiles? Explain with an example.
The treatment of alkyl chlorides with aqueous KOH leads to the formation of alcohols but in the presence of alcoholic KOH, alkenes are major products. Explain.
Identify 'A' in the following reaction -

(a) 2- Bromo-2 methylbutane
(b) 1 -Bromo-2,2-dimethylpropane
(c) 1 - Bromo - 3 -methylbutane
(d) 1 - Bromo- 2 -methylpropane
AgCN reacts with haloalkanes to form isocyanide. Haloalkanes react with KCN to form alkyl cyanides as the main product. Why?
Arrange the following organic compounds in descending order of their reactivity towards SN1 reaction.
C6H5CH2Br, C6H5CH(C6H5)Br, C6H5CH(CH3)Br, C6H5C(CH3)(C6H5)Br
Which of the following is optically inactive?
Which of the following pairs is/are correctly matched?
| Reaction | Product | |
| I | RX + AgCN | RNC |
| II | RX + KCN | RCN |
| III | RX + KNO2 | \[\begin{array}{cc} \phantom{.......}\ce{O}\\ \phantom{.....}/\\ \ce{R - N}\phantom{....}\\ \phantom{.....}\backslash\backslash\\ \phantom{.......}\ce{O} \end{array}\] |
| IV | RX + AgNO2 | \[\ce{R-O-N=O}\] |
Which of the following is a chiral compound?
Identify the end product (C) in the following sequence:
\[\ce{C2H5OH ->[SOCl2][Pyridine] A ->[KCN {(alc.)}] B ->[2H2O/H^+] C}\]
The increasing order of nucleophilicity would be:
How do polar solvents help in the first step in SN1 mechanism?
Aryl halides are extremely less reactive towards nucleophilic substitution. Predict and explain the order of reactivity of the following compounds towards nucleophilic substitution:
| (I) |  |
| (II) |  |
| (III) |  |
When CH3CH2CHCl2 is treated NaNH2 product formed is:-
The number of chiral alcohol (s) with molecular formula C4H10O is ______.
The number of chiral carbons present in the molecule given below is ______.

Assertion (A) : Nucleophilic substitution of iodoethane is easier than chloroethane.
Reason (R) : Bond enthalpy of C-I bond is less than that of C-Cl bond.
Explain why Grignard reagents should be prepared under anhydrous conditions.
Which alkyl halide from the following pair would you expect to react more rapidly by an SN2 mechanism? Explain your answer.
\[\begin{array}{cc}\ce{CH3CHCH2CH2Br}\\|\phantom{.........}\\\ce{CH3}\phantom{......}\end{array}\] or \[\begin{array}{cc}\ce{CH3CH2CHCH2Br}\\\phantom{}|\\\phantom{...}\ce{CH3}\end{array}\]
