Advertisements
Advertisements
प्रश्न
Compound ‘A’ with molecular formula \[\ce{C4H9Br}\] is treated with aq. \[\ce{KOH}\] solution. The rate of this reaction depends upon the concentration of the compound ‘A’ only. When another optically active isomer ‘B’ of this compound was treated with aq. \[\ce{KOH}\] solution, the rate of reaction was found to be dependent on concentration of compound and \[\ce{KOH}\] both.
(i) Write down the structural formula of both compounds ‘A’ and ‘B’.
(ii) Out of these two compounds, which one will be converted to the product with inverted configuration.
Advertisements
उत्तर
(i) The molecular formulae of isomers of \[\ce{C4H9Br}\] are \[\ce{CH3}\]
\[\begin{array}{cc}
\ce{CH3}\phantom{......}\\
|\phantom{.........}\\
\ce{CH3 - C - Br}\phantom{...........}\\
|\phantom{.........}\\
\ce{\underset{2 - Bromo-2-methyl propane (A)}{CH3}}\phantom{......}
\end{array}\]
\[\begin{array}{cc}
\ce{CH3 - CH2 - CH - CH3}\\
\phantom{.....}|\phantom{}\\
\phantom{.......}\ce{\underset{2-Bromobutance (B)}{Br}}\phantom{}
\end{array}\]
Since the rate of reaction of compound ‘A’ \[\ce{(C4H9Br)}\] with aqueous \[\ce{KOH}\] depends upon the concentration of compound ‘A’ only, therefore, the reaction occurs by SN1 mechanism and compound ‘A’ is tertiary bromide i.e., 2-Bromo-2-methylpropane.
\[\ce{(CH3)3CBr + KOH(aq) –> (CH3)3COH + KBr}\]
Rate = \[\ce{A:[(CH3)3CBr]}\]
(ii) Since compound ‘B’ is optically active and is an isomer of compound ‘A’ \[\ce{(C4H9Br)}\], therefore, compound ‘B’ must be 2-Bromobutane. Since the rate of reaction of compound ‘B’ with aqueous \[\ce{KOH}\] depends upon the concentration of compound ‘B’ and \[\ce{KOH}\], therefore, the reaction occurs by SN2 mechanism and product of hydrolysis will have inverted configuration.
\[\begin{array}{cc}
\ce{CH3CH2CHCH3 + KOH -> CH3CH2CHCH3 + KBr}\\
\phantom{..}|\phantom{...............................}|\phantom{...}\\
\phantom{..}\ce{Br}\phantom{.............................}\ce{OH}\phantom{.}
\end{array}\]
Rate = 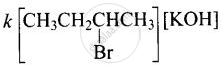
APPEARS IN
संबंधित प्रश्न
Out of  , which is more reactive towards SN1 reaction and why?
, which is more reactive towards SN1 reaction and why?
Which alkyl halide from the following pair would you expect to react more rapidly by an SN2 mechanism? Explain your answer.
CH3CH2CH2CH2Br or \[\begin{array}{cc}
\ce{CH3CH2CHCH3}\\
\phantom{...}|\\
\phantom{....}\ce{Br}\
\end{array}\]
In the following pair of halogen compounds, which compound undergoes a faster SN1 reaction?

Write the structure of the major organic product in the following reaction:
\[\ce{CH3CH2Br + KCN ->[aq.ethanol]}\]
C–Cl bond length in chlorobenzene is shorter than C–Cl bond length in CH3–Cl.
Given reasons: SN1 reactions are accompanied by racemization in optically active alkyl halides.
Answer the following question.
Write one stereochemical difference between SN1 and SN2 reactions.
In a coordination entity of the type [PtCl2(en)2]2+ which isomer will show optical isomerism?
Arrange the following organic compounds in descending order of their reactivity towards SN1 reaction.
C6H5CH2Br, C6H5CH(C6H5)Br, C6H5CH(CH3)Br, C6H5C(CH3)(C6H5)Br
Which of the following pairs is/are correctly matched?
| Reaction | Product | |
| I | RX + AgCN | RNC |
| II | RX + KCN | RCN |
| III | RX + KNO2 | \[\begin{array}{cc} \phantom{.......}\ce{O}\\ \phantom{.....}/\\ \ce{R - N}\phantom{....}\\ \phantom{.....}\backslash\backslash\\ \phantom{.......}\ce{O} \end{array}\] |
| IV | RX + AgNO2 | \[\ce{R-O-N=O}\] |
Which among MeX, RCH2X, R2CHX and R3CX is most reactive towards SN2 reaction?
The increasing order of nucleophilicity would be:
Which of the following compound will undergo racemisation when reacts with aq. KOH?
(i)
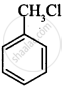
(ii)
CH3CH2CH2Cl
(iii)
\[\begin{array}{cc}
\ce{CH3}\phantom{..}\\
|\phantom{....}\\
\ce{CH3-CH-CH2Cl}
\end{array}\]
(iv)
\[\begin{array}{cc}
\phantom{..}\ce{H}\\
\phantom{..}|\\
\ce{CH3-C-Cl}\\
\phantom{..}|\\
\phantom{.....}\ce{C2H5}
\end{array}\]
Ethylene chloride and ethylidene chloride are isomers. Identify the correct statements.
(i) Both the compounds form same product on treatment with alcoholic KOH.
(ii) Both the compounds form same product on treatment with aq.NaOH.
(iii) Both the compounds form same product on reduction.
(iv) Both the compounds are optically active.
Why are aryl halides less reactive towards nucleophilic substitution reactions than alkyl halides?
The major product formed in the following reaction is:

Arrange the following compounds in increasing order of reactivity towards SN2 reaction.
2-Bromopentane, 1-Bromopentane, 2-Bromo-2-methylbutane
Inversion of configuration occurs in ______.
The following questions are case-based questions. Read the passage carefully and answer the questions that follow:
|
Nucleophilic Substitution: Influences of solvent polarity: The reaction rate (SN2) of 2-bromopropane and NaOH in ethanol containing 40% water is twice slower than in absolute ethanol. Hence the level of solvent polarity has an influence on both SN1 and SN2 reactions but with different results. Generally speaking, a weak polar solvent is favourable for SN2 reaction, while a strong polar solvent is favourable for SN1. Generally speaking, the substitution reaction of tertiary haloalkane is based on SN1 mechanism in solvents with a strong polarity (for example ethanol containing water). |
Answer the following questions:
(a) Why racemisation occurs in SN1? (1)
(b) Why is ethanol less polar than water? (1)
(c) Which one of, the following in each pair is more reactive towards SN2 reaction? (2)
(i) CH3 – CH2 – I or CH3CH2 – Cl
(ii)
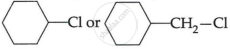
OR
(c) Arrange the following in the increasing order of their reactivity towards SN1 reactions: (2)
(i) 2-Bromo-2-methylbutane, 1-Bromo-pentane, 2-Bromo-pentane
(ii) 1-Bromo-3-methylbutane, 2-Bromo-2-methylbutane, 2-Bromo-3- methylbutane
Discuss the mechanism of alkaline hydrolysis of methyl bromide.
