Advertisements
Advertisements
प्रश्न
Why are aryl halides less reactive towards nucleophilic substitution reactions than alkyl halides?
Why are aryl halides less reactive than alkyl halides towards nucleophilic substitution reactions?
Why are haloarenes less reactive towards nucleophilic substitution reactions as compared to haloalkanes?
Advertisements
उत्तर
Aryl halides are less reactive towards nucleophilic substitution reaction due to the following reasons.
- In haloarenes, the lone pair of electron on halogen are in resonance with benzene ring. So, C – Cl bond acquires partial double bond character which strengthen C – Cl bond and difficult to be substituted by nucleophile.
Therefore, they are less reactive towards nucleophilic substitution reaction.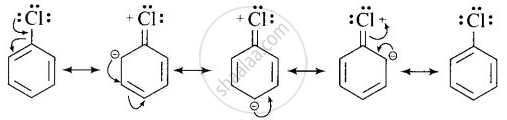
- In haloarenes, the carbon atom attached to halogen is sp2 hybridised. The sp2 hybridised carbon is more electronegative than sp3 hybridised carbon. This sp2-hybridised carbon in haloarenes can hold the electron pair of \[\ce{C - X}\] bond more tightly and make this \[\ce{C - Cl}\] bond shorter than \[\ce{C Cl}\] bond of haloalkanes.
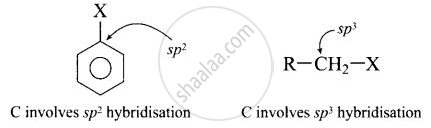
Since, it is difficult to break a shorter bond than a longer bond, therefore, halorenes are less reactive than haloalkanes. - In haloarenesm the phenyl cation is not stabilised by resonance therefore SN1 mechanism cannot be followed.
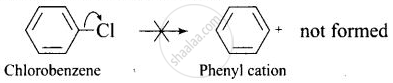
- Because of the repulsion between the nucleophile and electron-rich arenas, aryl halides are less reactive than alkyl halides.
संबंधित प्रश्न
Write the major products(s) in the following:

Which would undergo SN1 reaction faster in the following pair and why?

Identify 'A' in the following reaction -

(a) 2- Bromo-2 methylbutane
(b) 1 -Bromo-2,2-dimethylpropane
(c) 1 - Bromo - 3 -methylbutane
(d) 1 - Bromo- 2 -methylpropane
What is the action of the following on ethyl bromide?
silver acetate
Answer the following question.
Write one stereochemical difference between SN1 and SN2 reactions.
In a coordination entity of the type [PtCl2(en)2]2+ which isomer will show optical isomerism?
In the reaction, \[\ce{R - X + NaOR' -> ROR’ + X}\] ( – ve ion). The main product formed is:
Arrange the following organic compounds in descending order of their reactivity towards SN1 reaction.
C6H5CH2Br, C6H5CH(C6H5)Br, C6H5CH(CH3)Br, C6H5C(CH3)(C6H5)Br
Which of the following pairs is/are correctly matched?
| Reaction | Product | |
| I | RX + AgCN | RNC |
| II | RX + KCN | RCN |
| III | RX + KNO2 | \[\begin{array}{cc} \phantom{.......}\ce{O}\\ \phantom{.....}/\\ \ce{R - N}\phantom{....}\\ \phantom{.....}\backslash\backslash\\ \phantom{.......}\ce{O} \end{array}\] |
| IV | RX + AgNO2 | \[\ce{R-O-N=O}\] |
Which of the following compounds is optically active?
2-Bromopentane is heated with potassium ethoxide in ethanol. The major product obtained is ____________.
Among the following, the dissociation constant is highest for:
Which of the following alkyl halides will undergo SN1 reaction most readily?
Compound ‘A’ with molecular formula \[\ce{C4H9Br}\] is treated with aq. \[\ce{KOH}\] solution. The rate of this reaction depends upon the concentration of the compound ‘A’ only. When another optically active isomer ‘B’ of this compound was treated with aq. \[\ce{KOH}\] solution, the rate of reaction was found to be dependent on concentration of compound and \[\ce{KOH}\] both.
(i) Write down the structural formula of both compounds ‘A’ and ‘B’.
(ii) Out of these two compounds, which one will be converted to the product with inverted configuration.
How do polar solvents help in the first step in SN1 mechanism?
The number of chiral alcohol (s) with molecular formula C4H10O is ______.
The number of chiral carbons present in the molecule given below is ______.

Assertion (A) : Nucleophilic substitution of iodoethane is easier than chloroethane.
Reason (R) : Bond enthalpy of C-I bond is less than that of C-Cl bond.
