An SN1 reaction occurs through the formation of a carbocation intermediate. Alkyl halide (I) is tertiary (3°), whereas (II) is secondary (2°). As a result, (I) forms a tertiary carbocation, while (II) forms a secondary carbocation. Since the rate of an SN1 reaction increases with carbocation stability, and a tertiary carbocation is more stable than a secondary carbocation, compound (I), i.e., 2-chloro-2-methylpropane, undergoes the SN1 reaction faster than compound (II), i.e., 3-chloropentane.
Advertisements
Advertisements
प्रश्न
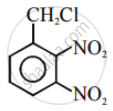
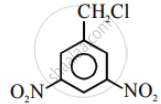
In the following pair of halogen compounds, which compound undergoes a faster SN1 reaction?

In the following pair of halogen compounds, which compound undergoes SN1 reaction faster and why?
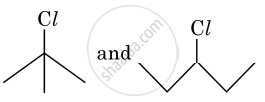
Advertisements
उत्तर १
The activity of halogen compounds in SN1 reaction depends on the stability of the carbocation formed due to ionization. The order of stability is tertiary > secondary > primary. Hence, 3° alkyl chloride is more active than 2° alkyl chloride. Hence, 2° alkyl chloride is more active in SN1 reaction.
 will react faster. The carbocation
will react faster. The carbocation 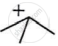 will be more stable and hence the reaction will be faster.
will be more stable and hence the reaction will be faster.
उत्तर २
Notes
Students should refer to the answer according to their questions.
APPEARS IN
संबंधित प्रश्न
Write the structure of an isomer of compound C4H9Br which is most reactive towards SN1 reaction
Write the major products(s) in the following:

Write the main products when methyl chloride is treated with AgCN.
Give reasons for the following:
(CH3)3C–O–CH3 on reaction with HI gives (CH3)3C–I and CH3–OH as the main products and not (CH3)3C–OH and CH3–I.
In the following pair of halogen compounds, which compound undergoes a faster SN1 reaction?

C–Cl bond length in chlorobenzene is shorter than C–Cl bond length in CH3–Cl.
AgCN reacts with haloalkanes to form isocyanide. Haloalkanes react with KCN to form alkyl cyanides as the main product. Why?
In the SN1 reaction, racemization takes place. It is due to:
SN1 reaction of alkyl halides lead to ___________.
Which of the following compounds will give a racemic mixture on nucleophilic substitution by OH ion?
1-Bromoethane, 1-Bromopropane, 1-Bromobutane, Bromobenzene
Complete the following analogy:
Same molecular formula but different structures: A : : Non superimposable mirror images: B
Which of the following statements are correct about this reaction?

(i) The given reaction follows SN2 mechanism.
(ii) (b) and (d) have opposite configuration.
(iii) (b) and (d) have same configuration.
(iv) The given reaction follows SN1 mechanism.
Cyanide ion acts as an ambident nucleophile. From which end it acts as a stronger nucleophile in aqueous medium? Give reason for your answer.
When CH3CH2CHCl2 is treated NaNH2 product formed is:-
Give reason for the following:
The product formed during SN1 reaction is a racemic mixture.
In which reaction mechanism carbocation is formed?
Which of the following compounds will show retention in configuration on nucleophile substitution by OH− ion?
The number of chiral carbons present in the molecule given below is ______.

In SN1 reactions, the correct order of reactivity for the following compounds:
CH3Cl, CH3CH2Cl, (CH3)2CHCl and (CH3)3CCl is ______.
The decreasing order of reactivity of the following compounds towards nucleophilic substitution (SN2) is ______.