Advertisements
Advertisements
प्रश्न
Give reasons for the following:
(CH3)3C–O–CH3 on reaction with HI gives (CH3)3C–I and CH3–OH as the main products and not (CH3)3C–OH and CH3–I.
Advertisements
उत्तर १
Usually, iodide, being a big nucleophile, attacks on the group with low steric hindrance and the reaction proceeds by SN2 mechanism.
However, in this case, methanol, on leaving generates a tertiary carbocation, which is more stable. Hence, this reaction proceeds by SN1 mechanism and therefore, we get (CH3)3C-I and CH3-OH as the major products.

उत्तर २
CH3)3-C–O–CH3 is an ether with two different alkyl groups, of which (CH3)3-C-, a tertiary alkyl group, on reaction with hydrogen halide (HI) forms a tertiary halide. This occurs as the reaction is an SN1 reaction. The reaction involves the formation of a stable carbocation. If the ether has a primary alkyl group, then the reaction follows the SN2 mechanism.
APPEARS IN
संबंधित प्रश्न
Which would undergo SN1 reaction faster in the following pair and why?

Write the structure of the major product in each of the following reaction :

Write the isomers of the compound having the formula C4H9Br.
Arrange the compounds of the following set in order of reactivity towards SN2 displacement:
2-Bromo-2-methylbutane, 1-Bromopentane, 2-Bromopentane
SN1 reactions are accompanied by racemization in optically active alkyl halides.
In the SN1 reaction, racemization takes place. It is due to:
Which of the following is an optically active compound?
Assertion: KCN reacts with methyl chloride to give methyl isocyanide.
Reason: CN– is an ambident nucleophile.
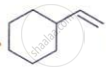 HCI, Major product ______.
HCI, Major product ______.
Which of the following reactions is an example of nucleophilic substitution reaction?
