Advertisements
Advertisements
प्रश्न
How the following conversion can be carried out?
Ethyl chloride to propanoic acid.
Write the chemical equation to convert the following:
Ethyl chloride to propanoic acid.
Advertisements
उत्तर
\[\ce{\underset{Ethyl chloride}{CH3CH2Cl} ->[KCN/EtOH-H2O][nucleophilic substitution] \underset{Propanenitrile}{CH3CH2CN} ->[H+/H2O][hydrolysis] \underset{Propanoic acid}{CH3CH2COOH}}\]
संबंधित प्रश्न
Give reasons for the following:
(CH3)3C–O–CH3 on reaction with HI gives (CH3)3C–I and CH3–OH as the main products and not (CH3)3C–OH and CH3–I.
Write the structure of the major product in each of the following reaction :

Write the structure of the major organic product in the following reaction:
\[\ce{CH3CH(Br)CH2CH3 + NaOH ->[water]}\]
Write the structure of the major organic product in the following reaction:
\[\ce{CH3CH2Br + KCN ->[aq{.} ethanol]}\]
Write the structure of the major organic product in the following reaction:
\[\ce{CH3CH2CH2OH + SOCl2 ->}\]
Write the mechanism of the following reaction:
\[\ce{{n}BuBr + KCN ->[EtOH-H2O] {n}BuCN}\]
The order of reactivities of the following alkyl halides for an SN2 reaction is:
Optically active isomers but not mirror images are called ____________.
Tertiary alkyl halides are practically inert to substitution by SN2 mechanism because of ____________.
Which of the following is the correct order of decreasing SN2 reactivity?
Which of the following is a chiral compound?
Identify the end product (C) in the following sequence:
\[\ce{C2H5OH ->[SOCl2][Pyridine] A ->[KCN {(alc.)}] B ->[2H2O/H^+] C}\]
Elimination reactions (especially β-elimination) are as common as the nucleophilic substitution reaction in case of alkyl halides. Specify the reagents used in both cases.
Cyanide ion acts as an ambident nucleophile. From which end it acts as a stronger nucleophile in aqueous medium? Give reason for your answer.
Match the reactions given in Column I with the types of reactions given in Column II.
| Column I | Column II | |
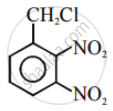
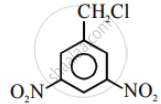
| (i) | 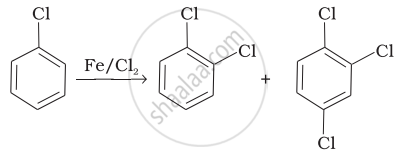 |
(a) Nucleophilic aromatic substitution |
| (ii) | \[\begin{array}{cc} \ce{CH3 - CH = CH2 + HBr -> CH3 - CH - CH3}\\ \phantom{............................}|\phantom{}\\ \phantom{.............................}\ce{Br}\phantom{} \end{array}\] |
(b) Electrophilic aromatic substitution |
| (iii) | 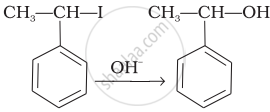 |
(c) Saytzeff elimination |
| (iv) | 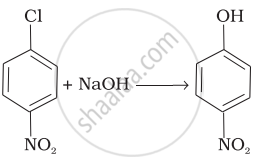 |
(d) Electrophilic addition |
| (v) | \[\begin{array}{cc} \ce{CH3 CH2 CH CH3 ->[alc.KOH] CH3 CH = CH CH3}\\ \phantom{}|\phantom{..........................}\\ \phantom{}\ce{Br}\phantom{........................} \end{array}\] |
(e) Nucleophilic substitution (SN1) |
The decreasing order of reactivity of the following compounds towards nucleophilic substitution (SN2) is ______.
Discuss the mechanism of alkaline hydrolysis of methyl bromide.
Explain why Grignard reagents should be prepared under anhydrous conditions.
Which of the following is halogen exchange reaction?
The correct order of increasing reactivity of
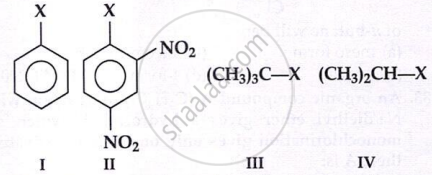
C-X bond towards nucleophile in the following compounds is: