Advertisements
Advertisements
प्रश्न
Discuss the mechanism of alkaline hydrolysis of bromomethane.
Advertisements
उत्तर
a. Alkaline hydrolysis of bromomethane follows bimolecular nucleophilic substitution (SN2)mechanism. The hydrolysis reaction can be written as follows:

1. Approach of the nucleophile (Backside attack):
i. The nucleophile (OH-) slowly approaches the carbon atom from the opposite side of the C - Br bond.
ii. C – OH weak bond is formed, while the existing C – Br bond gradually weakens.
iii. It is a slow process and hence, it is the rate determining step (R.D.S.).
2. Transition state (Activated complex): With the approach of OH- group and the
gradual departure of Br, a stage comes where the central atom is attached to five substituents. This state is known as transition state of reaction.
At this stage, the three hydrogen atoms lie in a plane perpendicular to the HO – C – Br axis.
3. Stereochemistry of SN2 reaction:
The attack might take place from back as well as from front side.
i. If backside attack takes place:
As shown in the figure given below, the OH group occupies a position in the product which is opposite to the position of Br. Similarly the positions of H2 and
H3 in the reactant and in the product are on opposite sides i.e., inverted due to the back side attack. This is known as inversion of configuration. Thus, backside
attack of nucleophile leads to inversion of configuration.

ii. If front side attack takes place: In this situation, the OH- occupies the same position which was occupied by Br in the reactant and the position of H1, H2 and
H3 also remain the same. Therefore, the configuration of the carbon is retained. This is known as retention of configuration.

The product X is obtained with inversion of configuration and not Y, with retention of configuration (X and Y are enantiomers). Thus, in SN2 reaction, the nucleophile attacks from backside leading to the inversion of configuration.
APPEARS IN
संबंधित प्रश्न
Write the structure of an isomer of compound C4H9Br which is most reactive towards SN1 reaction
How do you convert the following:
Ethanol to propanenitrile
Out of  , which is more reactive towards SN1 reaction and why?
, which is more reactive towards SN1 reaction and why?
Which would undergo SN2 reaction faster in the following pair and why ?

Write the isomers of the compound having the formula C4H9Br.
Arrange the compounds of the following set in order of reactivity towards SN2 displacement:
1-Bromo-3-methylbutane, 2-Bromo-2-methylbutane, 2-Bromo-3-methylbutane
What happens when chlorobenzene is subjected to hydrolysis?
What happens when methyl chloride is treated with KCN?
What is the action of the following on ethyl bromide
alcoholic solution of potassium hydroxide.
In a coordination entity of the type [PtCl2(en)2]2+ which isomer will show optical isomerism?
Arrange the following organic compounds in descending order of their reactivity towards SN1 reaction.
C6H5CH2Br, C6H5CH(C6H5)Br, C6H5CH(CH3)Br, C6H5C(CH3)(C6H5)Br
Which of the following is optically inactive?
Which of the following is an example of SN2 reaction?
The order of reactivities of the following alkyl halides for an SN2 reaction is:
SN2 mechanism proceeds through intervention of ____________.
Isopropyl chloride undergoes hydrolysis by:
Among the following, the dissociation constant is highest for:
Which of the following compounds will give a racemic mixture on nucleophilic substitution by OH ion?
1-Bromoethane, 1-Bromopropane, 1-Bromobutane, Bromobenzene

Which of the statements are correct about above reaction?
(i) (a) and (e) both are nucleophiles.
(ii) In (c) carbon atom is sp3 hybridised.
(iii) In (c) carbon atom is sp2 hybridised.
(iv) (a) and (e) both are electrophiles.
Which of the following is the definition of chirality?
Which one is the correct order of nucleophilic strength (pKa) of following nucleophiles?
Among the following compounds I - IV, which one forms a yellow precipitate on reacting sequentially with (i) NaOH (ii) dil. HNO3 (iii) AgNO3?
 |
 |
 |
 |
| I | II | III | IV |
Arrange the following compounds in increasing order of reactivity towards SN2 reaction.
2-Bromopentane, 1-Bromopentane, 2-Bromo-2-methylbutane
An organic compound A with the molecular formula (+) C4H9Br undergoes hydrolysis to form (+) C4H9OH. Give the structure of A and write the mechanism of the reaction.
Identify the product in the following reaction: 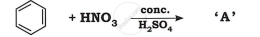
Which one of the following chlorohydrocarbons readily undergoes solvolysis?
Which of the following is halogen exchange reaction?
