An SN1 reaction occurs through the formation of a carbocation intermediate. Alkyl halide (I) is tertiary (3°), whereas (II) is secondary (2°). As a result, (I) forms a tertiary carbocation, while (II) forms a secondary carbocation. Since the rate of an SN1 reaction increases with carbocation stability, and a tertiary carbocation is more stable than a secondary carbocation, compound (I), i.e., 2-chloro-2-methylpropane, undergoes the SN1 reaction faster than compound (II), i.e., 3-chloropentane.
Advertisements
Advertisements
Questions
In the following pair of halogen compounds, which compound undergoes a faster SN1 reaction?

In the following pair of halogen compounds, which compound undergoes SN1 reaction faster and why?
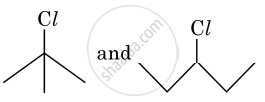
Advertisements
Solution 1
The activity of halogen compounds in SN1 reaction depends on the stability of the carbocation formed due to ionization. The order of stability is tertiary > secondary > primary. Hence, 3° alkyl chloride is more active than 2° alkyl chloride. Hence, 2° alkyl chloride is more active in SN1 reaction.
 will react faster. The carbocation
will react faster. The carbocation  will be more stable and hence the reaction will be faster.
will be more stable and hence the reaction will be faster.
Solution 2
Notes
Students should refer to the answer according to their questions.
APPEARS IN
RELATED QUESTIONS
How do you convert the following:
Ethanol to propanenitrile
In the following pair of halogen compounds, which compound undergoes a faster SN1 reaction?

Write the isomers of the compound having the formula C4H9Br.
Write the structure of the major organic product in the following reaction:
\[\ce{CH3CH(Br)CH2CH3 + NaOH ->[water]}\]
Write the structure of the major organic product in the following reaction:
\[\ce{CH3CH2CH2OH + SOCl2 ->}\]
C–Cl bond length in chlorobenzene is shorter than C–Cl bond length in CH3–Cl.
What is the action of the following on ethyl bromide:
moist silver oxide
What is the action of the following on ethyl bromide?
silver acetate
In the reaction, \[\ce{R - X + NaOR' -> ROR’ + X}\] ( – ve ion). The main product formed is:
Which of the following is the correct order of decreasing SN2 reactivity?
2-Bromopentane is heated with potassium ethoxide in ethanol. The major product obtained is ____________.
An organic molecule necessarily shows optical activity if it ____________.
The correct order of increasing the reactivity of C–X bond towards nucleophile in following compounds.

(I)

(II)
(CH3)3CCl
(III)
(CH3)2CHCl
(IV)
Which of the following compound will undergo racemisation when reacts with aq. KOH?
(i)
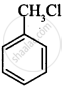
(ii)
CH3CH2CH2Cl
(iii)
\[\begin{array}{cc}
\ce{CH3}\phantom{..}\\
|\phantom{....}\\
\ce{CH3-CH-CH2Cl}
\end{array}\]
(iv)
\[\begin{array}{cc}
\phantom{..}\ce{H}\\
\phantom{..}|\\
\ce{CH3-C-Cl}\\
\phantom{..}|\\
\phantom{.....}\ce{C2H5}
\end{array}\]
Cyanide ion acts as an ambident nucleophile. From which end it acts as a stronger nucleophile in aqueous medium? Give reason for your answer.
The major product formed in the following reaction is:

The compound that will undergo SN1 reaction with the fastest rate is:
