Advertisements
Advertisements
Question
C–Cl bond length in chlorobenzene is shorter than C–Cl bond length in CH3–Cl.
Advertisements
Solution
In chlorobenzene, the C−Cl bond gets partial double bond character because of the +M effect shown by the Cl group. Due to double bond character, the bond length decreases in chlorobenzene as compared to the normal CH3−Cl bond length. Therefore, the C–Cl bond length in chlorobenzene is shorter than the C–Cl bond length in CH3–Cl.
APPEARS IN
RELATED QUESTIONS
Which would undergo SN1 reaction faster in the following pair and why?

Write the main products when methyl chloride is treated with AgCN.
Arrange the compounds of the following set in order of reactivity towards SN2 displacement:
1-Bromobutane, 1-Bromo-2, 2-dimethylpropane, 1-Bromo-2-methylbutane, 1-Bromo-3-methylbutane
The stability order for carbocation is _______.
(A) 2° > 3° > 1°
(B) 3° > 2° > 1°
(C) 3° > 1° > 2°
(D) 1° > 3° > 2°
In the SN1 reaction, racemization takes place. It is due to:
Among the following, the dissociation constant is highest for:
Identify the end product (C) in the following sequence:
\[\ce{C2H5OH ->[SOCl2][Pyridine] A ->[KCN {(alc.)}] B ->[2H2O/H^+] C}\]
Chlorination of alkanes is an example of
Which of the following is the definition of chirality?
The correct order of increasing reactivity of
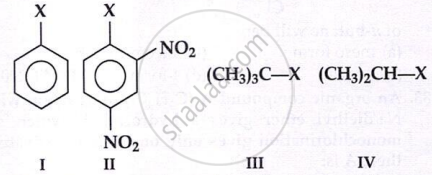
C-X bond towards nucleophile in the following compounds is:
