Advertisements
Advertisements
Questions
Give reasons:
The dipole moment of chlorobenzene is lower than that of cyclohexyl chloride.
Explain why the dipole moment of chlorobenzene is lower than that of cyclohexyl chloride?
Advertisements
Solution
- To understand the lower dipole moment of chlorobenzene, we must examine the contributing structures of the molecules.

- The C–Cl bond in chlorobenzene exhibits partial double-bond character (structures II, III, and IV). As a result, the C–Cl bond length is shorter than a single bond but longer than a double bond.
- The positive charge on the Cl atom minimises the expected negative (δ−) charge due to electronegativity.
- As a result, the dipole moment is determined by bond length and the partial negative charge on the Cl atom, which decreases. However, this does not occur with cyclohexyl chloride. The carbon in this alkyl halide is purely sp3 hybridised, with a bond length of a single bond and a greater dipole moment due to the presence of (δ−) on Cl.
APPEARS IN
RELATED QUESTIONS
What happens when \[\ce{CH3 - Br}\] is treated with KCN?
The presence of nitro group (−NO2) at o/p positions increases the reactivity of haloarenes towards nucleophilic substitution reactions.
How the following conversion can be carried out?
Chlorobenzene to p-nitrophenol
Write the product formed on reaction of D-glucose with Br2 water.
What is Grignard reagent?
Out of (CH3)3 C-Br and (CH3)3 C-I, which one is more reactive towards SN1 and why?
Assertion: Presence of a nitro group at ortho or para position increases the reactivity of haloarenes towards nucleophilic substitution.
Reason: Nitro group, being an electron-withdrawing group decreases the electron density over the benzene ring.
Arrange the following compounds in increasing order of rate of reaction towards nucleophilic substitution.
| (a) |  |
| (b) | 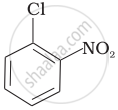 |
| (c) |  |
Arrange the following compounds in increasing order of rate of reaction towards nucleophilic substitution.
| (a) |  |
| (b) |  |
| (c) |  |
Arrange the following compounds in increasing order of rate of reaction towards nucleophilic substitution.
| (a) |  |
| (b) |  |
| (c) |  |
Arrange the following compounds in increasing order of rate of reaction towards nucleophilic substitution.
| (a) | 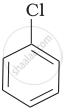 |
| (b) | 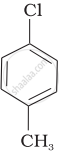 |
| (c) |  |
Haloarenes are less reactive than haloalkanes and haloalkenes. Explain.
\[\ce{C6H12O6 ->[(Zymase)] A ->[NaOH][\Delta] B + CHI3}\]
The number of carbon atoms present in the product B is:
In the reaction, \[\mathrm{CH}_3\mathrm{C}\equiv\mathrm{\overline{C}Na}^++(\mathrm{CH}_3)_2\mathrm{CHCl}\to\] the product formed is ______.
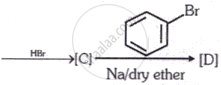
Identify the final product [D] obtained in the following sequence of reactions.
\[\ce{CH3CHO \underset{ii) H2O+}{\overset{i) LiAlH4}{->}} [A] \underset{\triangle}{\overset{H2SO4}{->}} [B]}\]