Advertisements
Advertisements
Question
What happens when \[\ce{CH3 - Br}\] is treated with KCN?
Advertisements
Solution
It is a nucleophilic substitution reaction. The nucleophile CN− substitutes Br−.
The reaction is as follows:
\[\ce{CH3BrKCN -> CH3CN + K Br}\]
APPEARS IN
RELATED QUESTIONS
Chlorobenzene is extremely less reactive towards a nucleophilic substitution reaction. Give two reasons for the same.
Write the product formed on reaction of D-glucose with Br2 water.
What is Grignard reagent?
Arrange the following compounds in increasing order of rate of reaction towards nucleophilic substitution.
| (a) |  |
| (b) |  |
| (c) |  |
Arrange the following compounds in increasing order of rate of reaction towards nucleophilic substitution.
| (a) | 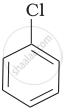 |
| (b) | 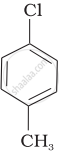 |
| (c) |  |
Allyl chloride is hydrolysed more readily than n-propyl chloride. Why?
\[\ce{C6H12O6 ->[(Zymase)] A ->[NaOH][\Delta] B + CHI3}\]
The number of carbon atoms present in the product B is:
Assertion: Chlorobenzene is resistant to nucleophilic substitution reaction at room temperature.
Reason (R): C–Cl bond gets weaker due, to resonance.
Why haloarenes are not reactive towards nucleophilic substitution reaction? Give two reactions.
In the reaction, \[\mathrm{CH}_3\mathrm{C}\equiv\mathrm{\overline{C}Na}^++(\mathrm{CH}_3)_2\mathrm{CHCl}\to\] the product formed is ______.
