Advertisements
Advertisements
Question
Arrange the following compounds in increasing order of rate of reaction towards nucleophilic substitution.
| (a) |  |
| (b) |  |
| (c) |  |
Options
(c) < (b) < (a)
(b) < (c) < (a)
(a) < (c) < (b)
(a) < (b) < (c)
Advertisements
Solution
(a) < (b) < (c)
Explanation:
The presence of an electron-withdrawing group (–NO2) at ortho- and para-positions increases the reactivity of haloarenes. The presence of a nitro group at ortho- and para-positions withdraws the electron density from the benzene ring and thus facilitates the attack of the nucleophile on haloarene. The carbanion thus formed is stabilised through resonance. The negative charge appeared at ortho- and para-positions with respect to the halogen substituent is stabilised by the –NO2 group.
APPEARS IN
RELATED QUESTIONS
What happens when \[\ce{CH3 - Br}\] is treated with KCN?
Chlorobenzene is extremely less reactive towards a nucleophilic substitution reaction. Give two reasons for the same.
Write the final product(s) in each of the following reactions:

The presence of nitro group (−NO2) at o/p positions increases the reactivity of haloarenes towards nucleophilic substitution reactions.
How the following conversion can be carried out?
Chlorobenzene to p-nitrophenol
Give reasons:
The dipole moment of chlorobenzene is lower than that of cyclohexyl chloride.
Assertion: Presence of a nitro group at ortho or para position increases the reactivity of haloarenes towards nucleophilic substitution.
Reason: Nitro group, being an electron-withdrawing group decreases the electron density over the benzene ring.
Which of the following compounds will give racemic mixture on nucleophilic substitution by \[\ce{OH-}\] ion?
(a) \[\begin{array}{cc}
\phantom{}\ce{CH3 - CH - Br}\\
\phantom{}|\\
\phantom{....}\ce{C2H5}\phantom{}
\end{array}\]
(b) \[\begin{array}{cc}
\phantom{..}\ce{Br}\\
\phantom{}|\\
\phantom{}\ce{CH3 - C - CH3}\\
\phantom{}|\\
\phantom{....}\ce{C2H5}\phantom{}
\end{array}\]
(c) \[\begin{array}{cc}
\phantom{....}\ce{CH3 - CH - CH2Br}\\
\phantom{}|\\
\phantom{....}\ce{C2H5}\phantom{}
\end{array}\]
Arrange the following compounds in increasing order of rate of reaction towards nucleophilic substitution.
| (a) |  |
| (b) | 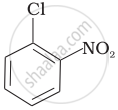 |
| (c) |  |
Arrange the following compounds in increasing order of rate of reaction towards nucleophilic substitution.
| (a) | 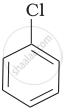 |
| (b) | 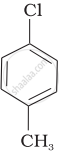 |
| (c) |  |
Haloarenes are less reactive than haloalkanes and haloalkenes. Explain.
Allyl chloride is hydrolysed more readily than n-propyl chloride. Why?
\[\ce{C6H12O6 ->[(Zymase)] A ->[NaOH][\Delta] B + CHI3}\]
The number of carbon atoms present in the product B is:
Assertion: Chlorobenzene is resistant to nucleophilic substitution reaction at room temperature.
Reason (R): C–Cl bond gets weaker due, to resonance.
Why haloarenes are not reactive towards nucleophilic substitution reaction? Give two reactions.
In the reaction, \[\mathrm{CH}_3\mathrm{C}\equiv\mathrm{\overline{C}Na}^++(\mathrm{CH}_3)_2\mathrm{CHCl}\to\] the product formed is ______.
