Advertisements
Advertisements
प्रश्न
C–Cl bond length in chlorobenzene is shorter than C–Cl bond length in CH3–Cl.
Advertisements
उत्तर
In chlorobenzene, the C−Cl bond gets partial double bond character because of the +M effect shown by the Cl group. Due to double bond character, the bond length decreases in chlorobenzene as compared to the normal CH3−Cl bond length. Therefore, the C–Cl bond length in chlorobenzene is shorter than the C–Cl bond length in CH3–Cl.
APPEARS IN
संबंधित प्रश्न
Discuss the mechanism of alkaline hydrolysis of bromomethane.
Which would undergo SN1 reaction faster in the following pair and why?

In the following pair of halogen compounds, which compound undergoes a faster SN1 reaction?

Arrange the compounds of the following set in order of reactivity towards SN2 displacement:
1-Bromo-3-methylbutane, 2-Bromo-2-methylbutane, 2-Bromo-3-methylbutane
Arrange the compounds of the following set in order of reactivity towards SN2 displacement:
1-Bromobutane, 1-Bromo-2, 2-dimethylpropane, 1-Bromo-2-methylbutane, 1-Bromo-3-methylbutane
The increasing order of nucleophilicity would be:
A primary alkyl halide would prefer to undergo ______.
Retention of configuration is observed in ______.
Give the mechanism of the following reaction:
\[\ce{CH3CH2OH ->[H2SO4][413 K] CH3CH2-O-CH2CH3 + H2O}\]
The following questions are case-based questions. Read the passage carefully and answer the questions that follow:
|
Nucleophilic Substitution: Influences of solvent polarity: The reaction rate (SN2) of 2-bromopropane and NaOH in ethanol containing 40% water is twice slower than in absolute ethanol. Hence the level of solvent polarity has an influence on both SN1 and SN2 reactions but with different results. Generally speaking, a weak polar solvent is favourable for SN2 reaction, while a strong polar solvent is favourable for SN1. Generally speaking, the substitution reaction of tertiary haloalkane is based on SN1 mechanism in solvents with a strong polarity (for example ethanol containing water). |
Answer the following questions:
(a) Why racemisation occurs in SN1? (1)
(b) Why is ethanol less polar than water? (1)
(c) Which one of, the following in each pair is more reactive towards SN2 reaction? (2)
(i) CH3 – CH2 – I or CH3CH2 – Cl
(ii)
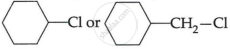
OR
(c) Arrange the following in the increasing order of their reactivity towards SN1 reactions: (2)
(i) 2-Bromo-2-methylbutane, 1-Bromo-pentane, 2-Bromo-pentane
(ii) 1-Bromo-3-methylbutane, 2-Bromo-2-methylbutane, 2-Bromo-3- methylbutane
