Advertisements
Advertisements
प्रश्न
The treatment of alkyl chlorides with aqueous KOH leads to the formation of alcohols but in the presence of alcoholic KOH, alkenes are major products. Explain.
Advertisements
उत्तर १
In an aqueous solution, KOH almost completely ionizes to give OH– ion, a strong nucleophile and reacts with alkyl halides to form alcohols. In an aqueous solution, OH– ions are highly hydrated. This reduces the basic character of OH– ions, due to which they fail to separate hydrogen atoms from the β-carbon of alkyl halide and cannot form an alkene.
On the other hand, alcoholic solution of KOH contains alkoxide (RO–) ions which, being a stronger base than OH–, easily remove HCl molecule from alkyl chloride to form alkene.
उत्तर २
Simple nucleophilic substitution occurs when alkyl chlorides react with aqueous KOH to form alcohols.
\[\ce{CH3 - CH2 - Cl + KOH ->[H2O]CH3 - CH2 - OH + KCl}\]
When aqueous KOH is substituted with alcoholic KOH, HCI is eliminated from an alkyl halide, resulting in the formation of alkenes instead of alcohols.
\[\ce{CH3 - CH2Cl + KOH ->[EtOH] CH2 = CH2}\]
This can be explained by the size of the nucleophile in both reactions. In an aqueous medium, the \[\ce{N\overset{\ominus}{u}}\] is \[\ce{\overset{\ominus}{O}H}\], which is small, whereas in an alcoholic medium, the \[\ce{N\overset{\ominus}{u}}\] is \[\ce{C2H^-5}\] is bulky.
The bulky \[\ce{N\overset{\ominus}{u}}\] always finds it easier to abstract a proton rather than attack a tetravalent carbon and form a substitution product.
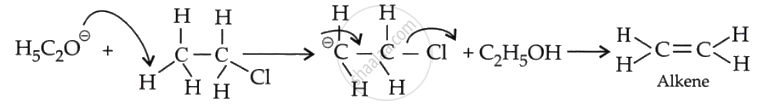
If C2H5OΘ attacks a carbon-carrying halogen, steric repulsions can delay the attack and prevent substitution.
APPEARS IN
संबंधित प्रश्न
Write the main products when methyl chloride is treated with AgCN.
Out of  , which is more reactive towards SN1 reaction and why?
, which is more reactive towards SN1 reaction and why?
Write the structures of A, B and C in the following:

Identify 'A' in the following reaction -

(a) 2- Bromo-2 methylbutane
(b) 1 -Bromo-2,2-dimethylpropane
(c) 1 - Bromo - 3 -methylbutane
(d) 1 - Bromo- 2 -methylpropane
AgCN reacts with haloalkanes to form isocyanide. Haloalkanes react with KCN to form alkyl cyanides as the main product. Why?
In the reaction, \[\ce{R - X + NaOR' -> ROR’ + X}\] ( – ve ion). The main product formed is:
Which compound in the following pair reacts faster in SN2 reaction with OH–?
- CH3Br or CH3
- CH3Cl, (CH3)3CCl
Arrange the following organic compounds in descending order of their reactivity towards SN1 reaction.
C6H5CH2Br, C6H5CH(C6H5)Br, C6H5CH(CH3)Br, C6H5C(CH3)(C6H5)Br
Which of the following is an example of SN2 reaction?
Which one is most reactive towards SN1 reaction?
The order of reactivity of the given haloalkanes towards nucleophile is:
Tertiary alkyl halides are practically inert to substitution by SN2 mechanism because of ____________.
Which of the following is a chiral compound?
Identify the end product (C) in the following sequence:
\[\ce{C2H5OH ->[SOCl2][Pyridine] A ->[KCN {(alc.)}] B ->[2H2O/H^+] C}\]
The reaction of C6H5–CH=CH–CH3 with HBr produces:
Compound ‘A’ with molecular formula \[\ce{C4H9Br}\] is treated with aq. \[\ce{KOH}\] solution. The rate of this reaction depends upon the concentration of the compound ‘A’ only. When another optically active isomer ‘B’ of this compound was treated with aq. \[\ce{KOH}\] solution, the rate of reaction was found to be dependent on concentration of compound and \[\ce{KOH}\] both.
(i) Write down the structural formula of both compounds ‘A’ and ‘B’.
(ii) Out of these two compounds, which one will be converted to the product with inverted configuration.
How do polar solvents help in the first step in SN1 mechanism?
When CH3CH2CHCl2 is treated NaNH2 product formed is:-
Discuss the mechanism of alkaline hydrolysis of methyl bromide.
Assertion (A):  undergoes SN2 reactions faster than
undergoes SN2 reactions faster than  .
.
Reason (R): Iodine is a better leaving group because of its large size.
In the light of the above statements, choose the correct answer from the options given below:
