Advertisements
Advertisements
Question
Write the structure of the major organic product in the following reaction:
\[\ce{CH3CH2CH2OH + SOCl2 ->}\]
Advertisements
Solution
\[\ce{\underset{Propan-1-ol}{CH3CH2CH2OH} + SOCl2 ->[nucleophilic substitution] \underset{1-Chloropropane}{CH3CH2CH2Cl} + HCl\uparrow + SO2\uparrow}\]
APPEARS IN
RELATED QUESTIONS
Discuss the mechanism of alkaline hydrolysis of bromomethane.
Write the structure of the major organic product in the following reaction:
\[\ce{CH3CH2Br + KCN ->[aq.ethanol]}\]
What happens when chlorobenzene is subjected to hydrolysis?
What happens when methyl chloride is treated with KCN?
What is the action of the following on ethyl bromide:
moist silver oxide
AgCN reacts with haloalkanes to form isocyanide. Haloalkanes react with KCN to form alkyl cyanides as the main product. Why?
Which of the following reactions is an example of nucleophilic substitution reaction?
Which among MeX, RCH2X, R2CHX and R3CX is most reactive towards SN2 reaction?
An important chemical method to resolve a racemic mixture makes use of the formation of ______.
The process of separation of a racemic modification into d and l-enantiomers is called ____________.
Which of the following compounds is optically active?
The increasing order of nucleophilicity would be:
Which of the following compound will undergo racemisation when reacts with aq. KOH?
(i)
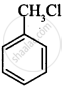
(ii)
CH3CH2CH2Cl
(iii)
\[\begin{array}{cc}
\ce{CH3}\phantom{..}\\
|\phantom{....}\\
\ce{CH3-CH-CH2Cl}
\end{array}\]
(iv)
\[\begin{array}{cc}
\phantom{..}\ce{H}\\
\phantom{..}|\\
\ce{CH3-C-Cl}\\
\phantom{..}|\\
\phantom{.....}\ce{C2H5}
\end{array}\]
A primary alkyl halide would prefer to undergo ______.
Which of the following is the definition of chirality?
The number of chiral carbons present in the molecule given below is ______.

Inversion of configuration occurs in ______.
Acetic anhydride from acetic acid
Explain why Grignard reagents should be prepared under anhydrous conditions.
