Advertisements
Advertisements
प्रश्न
Match the reactions given in Column I with the types of reactions given in Column II.
| Column I | Column II | |
| (i) | 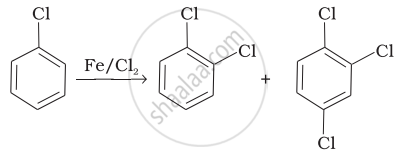 |
(a) Nucleophilic aromatic substitution |
| (ii) | \[\begin{array}{cc} \ce{CH3 - CH = CH2 + HBr -> CH3 - CH - CH3}\\ \phantom{............................}|\phantom{}\\ \phantom{.............................}\ce{Br}\phantom{} \end{array}\] |
(b) Electrophilic aromatic substitution |
| (iii) | 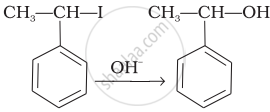 |
(c) Saytzeff elimination |
| (iv) | 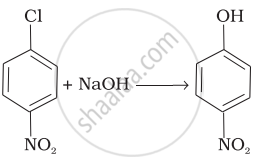 |
(d) Electrophilic addition |
| (v) | \[\begin{array}{cc} \ce{CH3 CH2 CH CH3 ->[alc.KOH] CH3 CH = CH CH3}\\ \phantom{}|\phantom{..........................}\\ \phantom{}\ce{Br}\phantom{........................} \end{array}\] |
(e) Nucleophilic substitution (SN1) |
Advertisements
उत्तर
| Column I | Column II | |
| (i) |  |
(b) Electrophilic aromatic substitution |
| (ii) | \[\begin{array}{cc} \ce{CH3 - CH = CH2 + HBr -> CH3 - CH - CH3}\\ \phantom{............................}|\phantom{}\\ \phantom{.............................}\ce{Br}\phantom{} \end{array}\] |
(d) Electrophilic addition |
| (iii) |  |
(e) Nucleophilic substitution (SN1) |
| (iv) |  |
(a) Nucleophilic aromatic substitution |
| (v) | \[\begin{array}{cc} \ce{CH3 CH2 CH CH3 ->[alc.KOH] CH3 CH = CH CH3}\\ \phantom{}|\phantom{..........................}\\ \phantom{}\ce{Br}\phantom{........................} \end{array}\] |
(c) Saytzeff elimination |
Explanation:
(i) In this reaction, an electrophile CF attacks on to the benzene ring and substitution takes place.
(ii) In this reaction, addition of \[\ce{HBr}\] takes place on to the doubly bonded carbons of propene in accordance with Markownikoff’s rule and electrophilic addition takes place.
(iii) In this reaction, the reactant is secondary halide. Here, halogen atom is substituted by hydroxyl ion. As it is secondary halide so it follows SN1 mechanism.
(iv) In this reaction, halogen atom is directly bonded to aromatic ring. So, it is nucleophilic aromatic substitution as OH– group has substituted halogen of given compound.
(v) It is an elimination reaction. It follows Saytzeff elimination rule.
APPEARS IN
संबंधित प्रश्न
How will you bring about the following conversion?
Toluene to benzyl alcohol
Out of C6H5CH2Cl and C6H5CHClC6H5, which is more easily hydrolysed by aqueous KOH.
Answer the following question.
Write one stereochemical difference between SN1 and SN2 reactions.
In a coordination entity of the type [PtCl2(en)2]2+ which isomer will show optical isomerism?
Which one is most reactive towards SN1 reaction?
In the SN1 reaction, racemization takes place. It is due to:
The order of reactivities of the following alkyl halides for an SN2 reaction is:
The process of separation of a racemic modification into d and l-enantiomers is called ____________.
Among the following, the dissociation constant is highest for:
A primary alkyl halide would prefer to undergo ______.

Which of the statements are correct about above reaction?
(i) (a) and (e) both are nucleophiles.
(ii) In (c) carbon atom is sp3 hybridised.
(iii) In (c) carbon atom is sp2 hybridised.
(iv) (a) and (e) both are electrophiles.
Aryl halides are extremely less reactive towards nucleophilic substitution. Predict and explain the order of reactivity of the following compounds towards nucleophilic substitution:
| (I) |  |
| (II) |  |
| (III) |  |
Cyanide ion acts as an ambident nucleophile. From which end it acts as a stronger nucleophile in aqueous medium? Give reason for your answer.
In which reaction mechanism carbocation is formed?
Which of the following compounds will show retention in configuration on nucleophile substitution by OH− ion?
Complete the reaction with the main product formed:

Convert bromoethane to propanamine.
Acetic anhydride from acetic acid
The compound that will undergo SN1 reaction with the fastest rate is:
