Advertisements
Advertisements
प्रश्न
Given reasons: C–Cl bond length in chlorobenzene is shorter than C–Cl bond length in CH3–Cl.
Advertisements
उत्तर
In chlorobenzene, the C−Cl bond gets partial double bond character because of the +M effect shown by the Cl group. Due to double bond character, the bond length decreases in chlorobenzene as compared to the normal CH3−Cl bond length. Therefore, the C–Cl bond length in chlorobenzene is shorter than the C–Cl bond length in CH3–Cl.
संबंधित प्रश्न
SN1 reactions are accompanied by racemization in optically active alkyl halides.
The stability order for carbocation is _______.
(A) 2° > 3° > 1°
(B) 3° > 2° > 1°
(C) 3° > 1° > 2°
(D) 1° > 3° > 2°
Given reasons: SN1 reactions are accompanied by racemization in optically active alkyl halides.
What is the action of the following on ethyl bromide
alcoholic solution of potassium hydroxide.
In a coordination entity of the type [PtCl2(en)2]2+ which isomer will show optical isomerism?
In the reaction, \[\ce{R - X + NaOR' -> ROR’ + X}\] ( – ve ion). The main product formed is:
Which of the following reactions is an example of nucleophilic substitution reaction?
Optically active isomers but not mirror images are called ____________.
Ethylene chloride and ethylidene chloride are isomers. Identify the correct statements.
(i) Both the compounds form same product on treatment with alcoholic KOH.
(ii) Both the compounds form same product on treatment with aq.NaOH.
(iii) Both the compounds form same product on reduction.
(iv) Both the compounds are optically active.
The correct order of increasing reactivity of
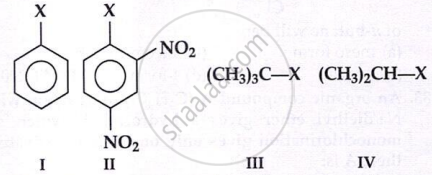
C-X bond towards nucleophile in the following compounds is:
