Advertisements
Advertisements
प्रश्न
Out of C6H5CH2Cl and C6H5CHClC6H5, which is more easily hydrolysed by aqueous KOH.
Advertisements
उत्तर
C6H5CHClC6H5 is hydrolysed faster.
- The hydrolysis of an alkyl halide is a nucleophilic substitution reaction. For aryl halides, this occurs through the SN1 method, resulting in a carbocation.
- C6H5CH2Cl or benzyl chloride gives
 a carbocation, while C6H5CHClC6H5 generates
a carbocation, while C6H5CHClC6H5 generates 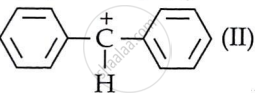 .
. - Out of I and II, carbocation II is more stable. Two attached phenyl rings on the carbon carry the positive charge.
- This leads to increased delocalisation of the positive charge and greater carbocation stability. This leads to faster formation of (II) and easier hydrolysis of the resulting halide than benzyl chloride.
APPEARS IN
संबंधित प्रश्न
Which would undergo SN1 reaction faster in the following pair and why?

Give reasons for the following:
(CH3)3C–O–CH3 on reaction with HI gives (CH3)3C–I and CH3–OH as the main products and not (CH3)3C–OH and CH3–I.
Out of  , which is more reactive towards SN1 reaction and why?
, which is more reactive towards SN1 reaction and why?
Write the isomers of the compound having the formula C4H9Br.
Arrange the compounds of the following set in order of reactivity towards SN2 displacement:
1-Bromobutane, 1-Bromo-2, 2-dimethylpropane, 1-Bromo-2-methylbutane, 1-Bromo-3-methylbutane
The treatment of alkyl chlorides with aqueous KOH leads to the formation of alcohols but in the presence of alcoholic KOH, alkenes are major products. Explain.
What happens when chlorobenzene is subjected to hydrolysis?
SN1 reactions are accompanied by racemization in optically active alkyl halides.
What is the action of the following on ethyl bromide
alcoholic solution of potassium hydroxide.
What is the action of the following on ethyl bromide?
silver acetate
In a coordination entity of the type [PtCl2(en)2]2+ which isomer will show optical isomerism?
Which one of the following halogen compounds is difficult to be hydrolysed by SN1 mechanism?
Which of the following compounds is optically active?
2-Bromopentane is heated with potassium ethoxide in ethanol. The major product obtained is ____________.
Which of the following compounds will give a racemic mixture on nucleophilic substitution by OH ion?
1-Bromoethane, 1-Bromopropane, 1-Bromobutane, Bromobenzene
A primary alkyl halide would prefer to undergo ______.
Cyanide ion acts as an ambident nucleophile. From which end it acts as a stronger nucleophile in aqueous medium? Give reason for your answer.
Inversion of configuration occurs in ______.
Convert bromoethane to propanamine.
