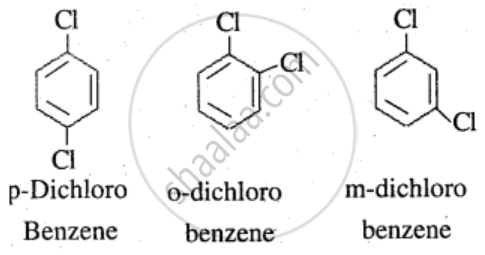
The melting point of p-dichlorobenzene is higher than that of its ortho- and meta-isomers due to its highly symmetrical structure. This symmetry allows the molecules to pack more efficiently and tightly in the crystal lattice, forming a more stable and orderly solid. As a result, stronger intermolecular forces hold the molecules together, and more energy (heat) is required to break these interactions during melting. In contrast, the less symmetrical o- and m-isomers pack less efficiently, have weaker intermolecular forces, and thus melt at lower temperatures.
Advertisements
Advertisements
प्रश्न
p-Dichlorobenzene has higher m.p. and lower solubility than those of o- and m-isomers. Discuss.
Why p-dichlorobenzene has higher melting point than those of ortho and meta-isomers?
p-Dichlorobenzene has higher m.p. than those of o- and m-isomers. Discuss.
Why is the melting point of p-dichlorobenzene higher than that of o- and m-isomers?
Advertisements
उत्तर
APPEARS IN
संबंधित प्रश्न
Give reasons : n-Butyl bromide has higher boiling point than t-butyl bromide.
Arrange the set of compounds in order of increasing boiling points.
Bromomethane, Bromoform, Chloromethane, Dibromomethane.
How many structural isomers are possible for a compound with the molecular formula C3H7Cl?
The decreasing order of boiling points of alkyl halides is:
Which of the following is liquid at room temperature (b.p. is shown against it)?
Which of the following possesses the highest melting point?
Mg reacts with RBr best in ____________.
p-dichlorobenzene has a higher melting point than its o- and m-isomers because ____________.
Which out of the following is an intensive property?
Why alkyl halides though polar are immiscible with water?
Why is the boiling point of o-dichlorobenzene higher than p-dichlorobenzene, but the melting point of para-isomer is higher than ortho-isomer?
Arrange the isomeric dichlorobenzene in the increasing order of their boiling point and melting points.
Write the structure of the following organic halogen compound.
4-tert-Butyl-3-iodoheptane
Write the structure of the following organic halogen compound.
4-tert-Butyl-3-iodoheptane
Write the structure of the following organic halogen compound.
4-tert-Butyl-3-iodoheptane
Write the structure of the following organic halogen compound:
4-tert-Butyl-3-iodoheptane
Write the structure of the following organic halogen compound.
4-tert-Butyl-3-iodoheptane
