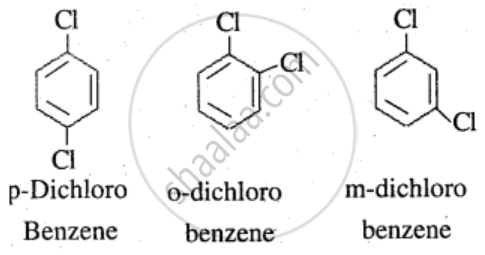
The melting point of p-dichlorobenzene is higher than that of its ortho- and meta-isomers due to its highly symmetrical structure. This symmetry allows the molecules to pack more efficiently and tightly in the crystal lattice, forming a more stable and orderly solid. As a result, stronger intermolecular forces hold the molecules together, and more energy (heat) is required to break these interactions during melting. In contrast, the less symmetrical o- and m-isomers pack less efficiently, have weaker intermolecular forces, and thus melt at lower temperatures.
Advertisements
Advertisements
प्रश्न
p-Dichlorobenzene has higher m.p. and lower solubility than those of o- and m-isomers. Discuss.
Why p-dichlorobenzene has higher melting point than those of ortho and meta-isomers?
p-Dichlorobenzene has higher m.p. than those of o- and m-isomers. Discuss.
Why is the melting point of p-dichlorobenzene higher than that of o- and m-isomers?
Advertisements
उत्तर
APPEARS IN
संबंधित प्रश्न
Arrange the set of compounds in order of increasing boiling points.
Bromomethane, Bromoform, Chloromethane, Dibromomethane.
Arrange the set of compounds in order of increasing boiling points.
1-Chloropropane, Isopropyl chloride, 1-Chlorobutane.
Why dextro and laevorotatory isomers of Butan-2-ol are difficult to separate by fractional distillation?
How many structural isomers are possible for a compound with the molecular formula C3H7Cl?
Which of the following halide is 2°?
Which of the following is liquid at room temperature (b.p. is shown against it)?
Which of the following possesses the highest melting point?
Mg reacts with RBr best in ____________.
p-dichlorobenzene has a higher melting point than its o- and m-isomers because ____________.
Reaction of \[\ce{C6H5CH2Br}\] with aqueous sodium hydroxide follows ______.
Arrange the following compounds in increasing order of their boiling points:
CH3CH2OH, CH3−CHO, CH3−COOH
Arrange the isomeric dichlorobenzene in the increasing order of their boiling point and melting points.
Write the structure of the following organic halogen compound.
1,4-Dibromobut-2-ene
Write the structure of the following organic halogen compound.
4-tert-Butyl-3-iodoheptane
Write the structure of the following organic halogen compound.
4-tert-Butyl-3-iodoheptane
Write the structure of the following organic halogen compound.
4-tert-Butyl-3-iodoheptane
Write the structure of the following organic halogen compound.
4-tert-Butyl-3-iodoheptane
