Advertisements
Advertisements
प्रश्न
Assertion: KCN reacts with methyl chloride to give methyl isocyanide.
Reason: CN– is an ambident nucleophile.
विकल्प
Assertion and reason both are correct and reason is correct explanation of assertion.
Assertion and reason both are wrong statements.
Assertion is correct but reason is wrong statement.
Assertion is wrong but reason is correct statement.
Assertion and reason both are correct statements but reason is not correct explanation of assertion.
Advertisements
उत्तर
Assertion is wrong but reason is correct statement.
Explanation:
\[\ce{\underset{Alkyl cyanide}{R - Cl + KCN –> R - CN + KCl}}\]
Isocyanide is not obtained in this reaction. CN– is an ambident nucleophile.
APPEARS IN
संबंधित प्रश्न
Which would undergo SN1 reaction faster in the following pair and why?

What is the action of the following on ethyl bromide?
silver acetate
Which of the following is an example of SN2 reaction?
Which one is most reactive towards SN1 reaction?
Most reactive halide towards SN1 reaction is ____________.
Optically active isomers but not mirror images are called ____________.
SN2 mechanism proceeds through intervention of ____________.
Which among MeX, RCH2X, R2CHX and R3CX is most reactive towards SN2 reaction?
2-Bromopentane is heated with potassium ethoxide in ethanol. The major product obtained is ____________.
Identify X and Y in the following sequence:
\[\ce{C2H5Br ->[X] Product ->[Y] C3H7NH2}\]
The increasing order of nucleophilicity would be:
Which of the following compound will undergo racemisation when reacts with aq. KOH?
(i)
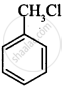
(ii)
CH3CH2CH2Cl
(iii)
\[\begin{array}{cc}
\ce{CH3}\phantom{..}\\
|\phantom{....}\\
\ce{CH3-CH-CH2Cl}
\end{array}\]
(iv)
\[\begin{array}{cc}
\phantom{..}\ce{H}\\
\phantom{..}|\\
\ce{CH3-C-Cl}\\
\phantom{..}|\\
\phantom{.....}\ce{C2H5}
\end{array}\]
Which of the following alkyl halides will undergo SN1 reaction most readily?
Ethylene chloride and ethylidene chloride are isomers. Identify the correct statements.
(i) Both the compounds form same product on treatment with alcoholic KOH.
(ii) Both the compounds form same product on treatment with aq.NaOH.
(iii) Both the compounds form same product on reduction.
(iv) Both the compounds are optically active.
Which of the compounds will react faster in SN1 reaction with the –OH ion?
\[\ce{CH3-CH2-Cl}\] or \[\ce{C6H5-CH2-Cl}\]
Why are aryl halides less reactive towards nucleophilic substitution reactions than alkyl halides?
CCl4 is insoluble in water because:-
Which one of the following compounds is more reactive towards SN1 reaction?
The major product formed in the following reaction is:

Which of the following compounds will show retention in configuration on nucleophile substitution by OH− ion?
