Advertisements
Advertisements
प्रश्न
What is the action of the following on ethyl bromide?
silver acetate
Write the chemical reaction for the following:
Ethyl bromide is heated with silver acetate.
Advertisements
उत्तर
\[\ce{\underset{\text{Ethyl bromide}}{C2H5Br} + \underset{\text{Silver acetate}}{CH3COOAg}->\underset{\text{Ethyl acetate}}{CH3COOC2H5} + AgBr}\]
संबंधित प्रश्न
Which would undergo SN1 reaction faster in the following pair and why?

Give reasons for the following:
(CH3)3C–O–CH3 on reaction with HI gives (CH3)3C–I and CH3–OH as the main products and not (CH3)3C–OH and CH3–I.
Given reasons: C–Cl bond length in chlorobenzene is shorter than C–Cl bond length in CH3–Cl.
Write the structure of the major organic product in the following reaction:
\[\ce{CH3CH2Br + KCN ->[aq.ethanol]}\]
Arrange the compounds of the following set in order of reactivity towards SN2 displacement:
1-Bromo-3-methylbutane, 2-Bromo-2-methylbutane, 2-Bromo-3-methylbutane
What happens when methyl chloride is treated with KCN?
C–Cl bond length in chlorobenzene is shorter than C–Cl bond length in CH3–Cl.
The stability order for carbocation is _______.
(A) 2° > 3° > 1°
(B) 3° > 2° > 1°
(C) 3° > 1° > 2°
(D) 1° > 3° > 2°
What is the action of the following on ethyl bromide
alcoholic solution of potassium hydroxide.
Which of the following is optically inactive?
Which of the following is a primary halide?
Optically active isomers but not mirror images are called ____________.
Isopropyl chloride undergoes hydrolysis by:
Which of the following is the correct order of decreasing SN2 reactivity?
Racemic compound has ____________.
Which of the following undergoes nucleophilic substitution exclusively by SN1 mechanism?
The correct order of increasing the reactivity of C–X bond towards nucleophile in following compounds.

(I)

(II)
(CH3)3CCl
(III)
(CH3)2CHCl
(IV)
The increasing order of reactivity towards SN1 mechanism is:
(I) \[\begin{array}{cc}
\ce{CH3-CH-CH2-CH3}\\
|\phantom{........}\\
\ce{CH3}\phantom{.....}
\end{array}\]
(II) CH3CH2CH2Cl
(III) P–CH3O–C6H4–CH2Cl
Which of the following compounds will give a racemic mixture on nucleophilic substitution by OH ion?
1-Bromoethane, 1-Bromopropane, 1-Bromobutane, Bromobenzene
Assertion: KCN reacts with methyl chloride to give methyl isocyanide.
Reason: CN– is an ambident nucleophile.

Which of the statements are correct about above reaction?
(i) (a) and (e) both are nucleophiles.
(ii) In (c) carbon atom is sp3 hybridised.
(iii) In (c) carbon atom is sp2 hybridised.
(iv) (a) and (e) both are electrophiles.
Which of the following statements are correct about this reaction?

(i) The given reaction follows SN2 mechanism.
(ii) (b) and (d) have opposite configuration.
(iii) (b) and (d) have same configuration.
(iv) The given reaction follows SN1 mechanism.
Write the structures and names of the compounds formed when compound ‘A’ with molecular formula, \[\ce{C7H8}\] is treated with \[\ce{Cl2}\] in the presence of \[\ce{FeCl3}\].
Match the reactions given in Column I with the types of reactions given in Column II.
| Column I | Column II | |
| (i) | 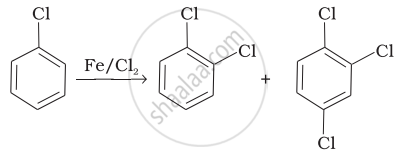 |
(a) Nucleophilic aromatic substitution |
| (ii) | \[\begin{array}{cc} \ce{CH3 - CH = CH2 + HBr -> CH3 - CH - CH3}\\ \phantom{............................}|\phantom{}\\ \phantom{.............................}\ce{Br}\phantom{} \end{array}\] |
(b) Electrophilic aromatic substitution |
| (iii) | 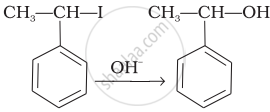 |
(c) Saytzeff elimination |
| (iv) | 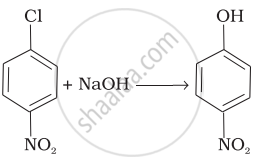 |
(d) Electrophilic addition |
| (v) | \[\begin{array}{cc} \ce{CH3 CH2 CH CH3 ->[alc.KOH] CH3 CH = CH CH3}\\ \phantom{}|\phantom{..........................}\\ \phantom{}\ce{Br}\phantom{........................} \end{array}\] |
(e) Nucleophilic substitution (SN1) |
Give reason for the following:
The product formed during SN1 reaction is a racemic mixture.
An organic compound A with the molecular formula (+) C4H9Br undergoes hydrolysis to form (+) C4H9OH. Give the structure of A and write the mechanism of the reaction.
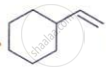 HCI, Major product ______.
HCI, Major product ______.
The correct order of increasing reactivity of
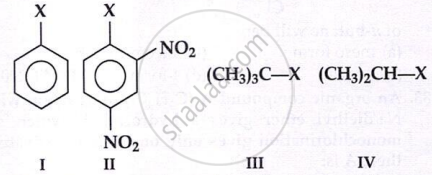
C-X bond towards nucleophile in the following compounds is:
