Advertisements
Advertisements
प्रश्न
Ethylene chloride and ethylidene chloride are isomers. Identify the correct statements.
(i) Both the compounds form same product on treatment with alcoholic KOH.
(ii) Both the compounds form same product on treatment with aq.NaOH.
(iii) Both the compounds form same product on reduction.
(iv) Both the compounds are optically active.
Advertisements
उत्तर
(i) Both the compounds form same product on treatment with alcoholic KOH.
(iii) Both the compounds form same product on reduction.
Explanation:
\[\ce{\underset{(ethylidence chloride)}{H3C - CHCl2}}\] and \[\begin{array}{cc}
\phantom{}\ce{H2C - CH2}\phantom{}\\
\phantom{}|\phantom{....}|\\
\phantom{.}\ce{\underset{(ethylene dichloride)}{\phantom{}Cl\phantom{...}Cl}}\phantom{}
\end{array}\] are isomers.
(i) They give ethyne on treatment with alcoholic KOH.
\[\ce{CH3CHCl2 ->[alc][KOH] CH ≡ CH + 2KCl + 2H2O}\]
\[\ce{Cl - CH2 - CH2 - Cl ->[alc.KOH] CH ≡ CH + 2KCl + H2O}\]
(ii) On reduction with Zn dust in alcohol they give ethylene.
\[\ce{CH3CHCl2 + Zn ->[CH3OH] CH2 ≡ CH}\]
\[\ce{Cl - CH2 - CH2 - Cl + Zn ->[CH3OH] CH2 = CH2}\]
APPEARS IN
संबंधित प्रश्न
Write the structures of A, B and C in the following:

Write the isomers of the compound having the formula C4H9Br.
Which compound in the following pair will react faster in SN2 reaction with OH−?
CH3Br or CH3I
How the following conversion can be carried out?
Ethyl chloride to propanoic acid.
Given reasons: SN1 reactions are accompanied by racemization in optically active alkyl halides.
What is the action of the following on ethyl bromide:
moist silver oxide
SN2 mechanism proceeds through intervention of ____________.
Which of the following is a chiral compound?
Which of the following compound will undergo racemisation when reacts with aq. KOH?
(i)
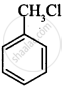
(ii)
CH3CH2CH2Cl
(iii)
\[\begin{array}{cc}
\ce{CH3}\phantom{..}\\
|\phantom{....}\\
\ce{CH3-CH-CH2Cl}
\end{array}\]
(iv)
\[\begin{array}{cc}
\phantom{..}\ce{H}\\
\phantom{..}|\\
\ce{CH3-C-Cl}\\
\phantom{..}|\\
\phantom{.....}\ce{C2H5}
\end{array}\]
The increasing order of reactivity towards SN1 mechanism is:
(I) \[\begin{array}{cc}
\ce{CH3-CH-CH2-CH3}\\
|\phantom{........}\\
\ce{CH3}\phantom{.....}
\end{array}\]
(II) CH3CH2CH2Cl
(III) P–CH3O–C6H4–CH2Cl
Which of the following compounds will give a racemic mixture on nucleophilic substitution by OH ion?
1-Bromoethane, 1-Bromopropane, 1-Bromobutane, Bromobenzene
A primary alkyl halide would prefer to undergo ______.

Which of the statements are correct about above reaction?
(i) (a) and (e) both are nucleophiles.
(ii) In (c) carbon atom is sp3 hybridised.
(iii) In (c) carbon atom is sp2 hybridised.
(iv) (a) and (e) both are electrophiles.
Which of the compounds will react faster in SN1 reaction with the –OH ion?
\[\ce{CH3-CH2-Cl}\] or \[\ce{C6H5-CH2-Cl}\]
Why are aryl halides less reactive towards nucleophilic substitution reactions than alkyl halides?
Chlorination of alkanes is an example of
Which one is the correct order of nucleophilic strength (pKa) of following nucleophiles?
Identify the product in the following reaction: 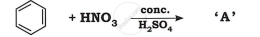
Discuss the mechanism of alkaline hydrolysis of methyl bromide.
Which of the following reactions is an example of nucleophilic substitution reaction?
