Advertisements
Advertisements
प्रश्न
Haloalkanes contain halogen atom (s) attached to the sp3 hybridised carbon atom of an alkyl group. Identify haloalkane from the following compounds.
(i) 2-Bromopentane
(ii) Vinyl chloride (chloroethene)
(iii) 2-chloroacetophenone
(iv) Trichloromethane
Advertisements
उत्तर
(i) 2-Bromopentane
(iv) Trichloromethane
Explanation:
\[\begin{array}{cc}
\ce{CH3CHCH2CH2CH3}\\
\phantom{}|\phantom{..........}\\
\phantom{}\ce{\underset{2-Bromopentane}{Br}}\phantom{.........}
\end{array}\]
\[\begin{array}{cc}
\ce{Cl - CH - Cl}\\
\phantom{}|\phantom{..}\\
\phantom{}\ce{\underset{Trichloromethane}{Cl}}\phantom{.}
\end{array}\]
In both of these compounds halogen atoms are attached to sp3 hybridised carbon atom of alkyl group.
APPEARS IN
संबंधित प्रश्न
Draw the structure of the major monohalo product in the following reaction:

Draw the structures of major monohalo products in each of the following reaction:

Name the following halide according to the IUPAC system and classify it as an alkyl, allyl, benzoyl (primary, secondary, tertiary), vinyl or aryl halide:
CH3CH = C(Cl)CH2CH(CH3)2
Name the following halide according to the IUPAC system and classify it as an alkyl, allyl, benzoyl (primary, secondary, tertiary), vinyl or aryl halide:
CH3CH = CHC(Br)(CH3)2
What should be the correct IUPAC name for diethylbromomethane?
The reaction of toluene with chlorine in the presence of iron and in the absence of light yields ______.
Name the following halides according to IUPAC system and classify them as alkyl, allyl, benzyl (primary, secondary, tertiary), vinyl or aryl halides:
\[\ce{CH3CH2C(CH3)2CH2I}\]
Name the following halides according to IUPAC system and classify them as alkyl, allyl, benzyl (primary, secondary, tertiary), vinyl or aryl halides:
\[\ce{CH3C(C2H5)2CH2Br}\]
Name the following halides according to the IUPAC system and classify them as alkyl, allyl, benzyl (primary, secondary, tertiary), vinyl or aryl halides:
\[\ce{CH3C(C2H5)2CH2Br}\]
Name the following halide according to the IUPAC system and classify it as alkyl, allyl, benzyl (primary, secondary, tertiary), vinyl or aryl halide:
CH3C(Cl)(C2H5)CH2CH3
Name the following halide according to IUPAC system and classify them as alkyl, allyl, benzyl (primary, secondary, tertiary), vinyl or aryl halides:
\[\ce{CH3CH2C(CH3)2CH2I}\]
Name the following halides according to IUPAC system and classify them as alkyl, allyl, benzyl (primary, secondary, tertiary), vinyl or aryl halides:
CH3C(Cl)(C2H5 )CH2CH3
Name the following halide according to IUPAC system and classify them as alkyl, allyl, benzyl (primary, secondary, tertiary), vinyl or aryl halides:
\[\ce{CH3C(C2H5)2CH2Br}\]
Name the following halide according to IUPAC system and classify as alkyl, allyl, benzyl (primary, secondary, tertiary), vinyl or aryl halide:
\[\ce{CH3C(C2H5)2CH2Br}\]
Draw the structure of the major monohalo product in the following reaction:

Which is the correct IUPAC name for 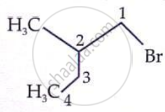 is______.
is______.
Which of the following is an example of vicdihalide?
Ethylidene chloride is a/an ______.
