Advertisements
Advertisements
Question
Haloalkanes contain halogen atom (s) attached to the sp3 hybridised carbon atom of an alkyl group. Identify haloalkane from the following compounds.
(i) 2-Bromopentane
(ii) Vinyl chloride (chloroethene)
(iii) 2-chloroacetophenone
(iv) Trichloromethane
Advertisements
Solution
(i) 2-Bromopentane
(iv) Trichloromethane
Explanation:
\[\begin{array}{cc}
\ce{CH3CHCH2CH2CH3}\\
\phantom{}|\phantom{..........}\\
\phantom{}\ce{\underset{2-Bromopentane}{Br}}\phantom{.........}
\end{array}\]
\[\begin{array}{cc}
\ce{Cl - CH - Cl}\\
\phantom{}|\phantom{..}\\
\phantom{}\ce{\underset{Trichloromethane}{Cl}}\phantom{.}
\end{array}\]
In both of these compounds halogen atoms are attached to sp3 hybridised carbon atom of alkyl group.
APPEARS IN
RELATED QUESTIONS
Draw the structure of the major monohalo product in the following reaction:

Name the following halide according to the IUPAC system and classify it as an alkyl, allyl, benzoyl (primary, secondary, tertiary), vinyl or aryl halide:
CH3C(Cl)(C2H5)CH2CH3
Name the following halide according to the IUPAC system and classify it as an alkyl, allyl, benzoyl (primary, secondary, tertiary), vinyl or aryl halide:
p-ClC6H4CH2CH(CH3)2
Draw the structure of major monohalo product in each of the following reactions :
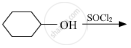
What should be the correct IUPAC name for diethylbromomethane?
Which of the carbon atoms present in the molecule given below are asymmetric?
\[\begin{array}{cc}
\phantom{}\ce{HO}\phantom{.....}\ce{OH}\phantom{..}\ce{H}\phantom{.....}\ce{O}\phantom{..}\\
\phantom{..}\backslash\phantom{.....}|\phantom{....}|\phantom{.....}//\phantom{.}\\
\ce{\overset{a}{C} - \overset{b}{C} - \overset{c}{C} - \overset{d}{C}}\\
\phantom{..}//\phantom{.....}|\phantom{....}|\phantom{....}\phantom{.}\backslash\phantom{...}\\
\phantom{}\ce{O}\phantom{......}\ce{H}\phantom{...}\ce{OH}\phantom{...}\ce{H}\phantom{}\\
\end{array}\]
Which of the products will be major product in the reaction given below? Explain.
\[\ce{CH3CH = CH2 + HI -> \underset{(A)}{CH3CH2CH2I} + \underset{(B)}{CH3CHICH3}}\]
Classify the following compound as a primary, secondary and tertiary halide.
2-Bromo-2-methylpropane
Match the items of Column I and Column II.
| Column I | Column II | |
| (i) | SN1 reaction | (a) vic-dibromides |
| (ii) | Chemicals in fire extinguisher | (b) gem-dihalides |
| (iii) | Bromination of alkenes | (c) Racemisation |
| (iv) | Alkylidene halides | (d) Saytzeff rule |
| (v) | Elimination of HX from alkylhalide | (e) Chlorobromocarbons |
Classify the following compound as a primary, secondary and tertiary halide.
1-Bromobut-2-ene
Which of the following belongs to the class of vinyl halides?
Name the following halides according to the IUPAC system and classify them as alkyl, allyl, benzyl (primary, secondary, tertiary), vinyl or aryl halides:
\[\ce{CH3C(C2H5)2CH2Br}\]
Name the following halide according to the IUPAC system and classify it as alkyl, allyl, benzyl (primary, secondary, tertiary), vinyl or aryl halide:
\[\ce{CH_3CH_2C(CH_3)_2CH_2I}\]
Name the following halide according to the IUPAC system and classify it as alkyl, allyl, benzyl (primary, secondary, tertiary), vinyl or aryl halide:
CH3CH2C(CH3)2CH2I
Name the following halide according to IUPAC system and classify them as alkyl, allyl, benzyl (primary, secondary, tertiary), vinyl or aryl halides:
\[\ce{CH3C(C2H5)2CH2Br}\]
Name the following halide according to IUPAC system and classify as alkyl, allyl, benzyl (primary, secondary, tertiary), vinyl or aryl halides:
CH3CH2C(CH3)2CH2l
Name the following halide according to IUPAC system and classify as alkyl, allyl, benzyl (primary, secondary, tertiary), vinyl or aryl halide:
\[\ce{CH3C(Cl)(C2H5)CH2CH3}\]
Name the following halide according to IUPAC system and classify as alkyl, allyl, benzyl (primary, secondary, tertiary), vinyl or aryl halide:
\[\ce{CH3C(C2H5)2CH2Br}\]
Name the following halide according to IUPAC system and classify them as alkyl, allyl, benzyl (primary, secondary, tertiary), vinyl or aryl halides:
\[\ce{CH3C(C2H5)2CH2Br}\]
The IUPAC name of the compound shown below is: