Advertisements
Advertisements
प्रश्न
A primary alkyl halide would prefer to undergo ______.
पर्याय
SN1 reaction
SN2 reaction
α–Elimination
Racemisation
Advertisements
उत्तर
A primary alkyl halide would prefer to undergo SN2 reaction.
Explanation:
SN2 type reactions (i.e. bimolecular nucleophilic substitution) proceed in one step and the rate of reaction depends on concentration of alkyl halide as well as nucleophile i.e. r = k[RX][Nu]. It is a second-order reaction. During SN2 reaction, inversion in configuration occurs (viz. starting with dextrorotatory halide a laevorotatory product is obtained and vice-versa).
APPEARS IN
संबंधित प्रश्न
Which would undergo SN2 reaction faster in the following pair and why ?

Write the structure of the major product in each of the following reaction :

Which compound in the following pair will react faster in SN2 reaction with OH−?
CH3Br or CH3I
What is the action of the following on ethyl bromide:
moist silver oxide
What is the action of the following on ethyl bromide?
moist silver oxide
Which of the following is an example of SN2 reaction?
Which of the following is a primary halide?
An important chemical method to resolve a racemic mixture makes use of the formation of ______.
The process of separation of a racemic modification into d and l-enantiomers is called ____________.
Which of the following compounds is optically active?
Which of the following undergoes nucleophilic substitution exclusively by SN1 mechanism?
Which of the following alkyl halides will undergo SN1 reaction most readily?
Complete the following analogy:
Same molecular formula but different structures: A : : Non superimposable mirror images: B
Which of the compounds will react faster in SN1 reaction with the –OH ion?
\[\ce{CH3-CH2-Cl}\] or \[\ce{C6H5-CH2-Cl}\]
Cyanide ion acts as an ambident nucleophile. From which end it acts as a stronger nucleophile in aqueous medium? Give reason for your answer.
Which of the following is the definition of chirality?
Which alkyl halide from the following pair would you expect to react more rapidly by an SN2 mechanism? Explain your answer.
\[\begin{array}{cc}
\ce{CH3CHCH2CH2Br}\\
|\phantom{.............}\\
\ce{CH3}\phantom{..........}\\
\end{array}\] or \[\begin{array}{cc}
\ce{CH3CH2CHCH2Br}\\
|\\
\phantom{...}\ce{CH3}\\
\end{array}\]
Which of the following is halogen exchange reaction?
The correct order of increasing reactivity of
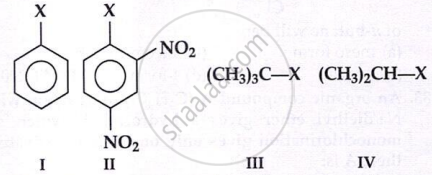
C-X bond towards nucleophile in the following compounds is:
