Advertisements
Advertisements
Question
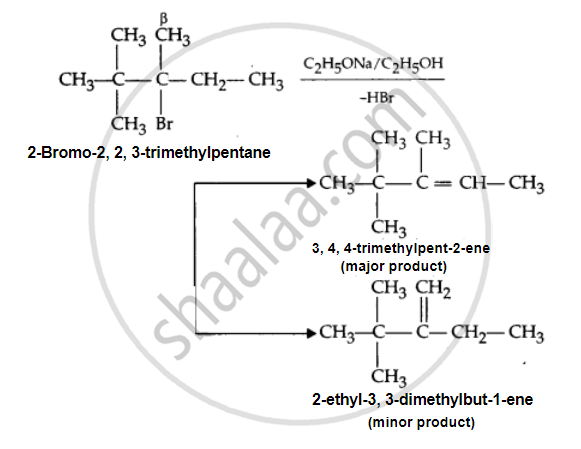
Predict all the alkenes that would be formed by dehydrohalogenation of the following halide with sodium ethoxide in ethanol and identify the major alkene:
2, 2, 3-Trimethyl-3-bromopentane
Advertisements
Solution
There are two different types of β-hydrogen atoms present in the halide. Hence, in the dehydrohalogenation reaction, it will form two alkenes: 3, 4, 4-trimethylpent-2-ene and 2-ethyl-3, 3-dimethylbut-1-ene. The first alkene is more stable because it is more substituted (according to the Saytzeff rule). Hence, it is the major product.
APPEARS IN
RELATED QUESTIONS
Identify the product ‘D’ in the following sequence of reactions:
\[\ce{H3C - CH2 - CH2 - Cl \underset{KOH}{\overset{Alc}{->}} 'B' \overset{HBr}{->} 'C' \underset{Elther}{\overset{Na}{->}}'D'}\]
Identify ‘A’ and ‘B’ in the following reaction :
\[\ce{CH3 - CH = CH2 ->[HBr]'A' ->[alc.KOH]'B'}\]
How do you convert:
2-bromobutane to but-2-ene
Write the main products when n-butyl chloride is treated with alcoholic KOH.
Predict all the alkenes that would be formed by dehydrohalogenation of the following halide with sodium ethoxide in ethanol and identify the major alkene:
1-Bromo-1-methylcyclohexane
Predict all the alkenes that would be formed by dehydrohalogenation of the following halide with sodium ethoxide in ethanol and identify the major alkene:
2-Chloro-2-methylbutane
How the following conversion can be carried out?
1-Bromopropane to 2-bromopropane
How the following conversion can be carried out?
2-Chloropropane to 1-propanol

'A' is:
Deamination of meso- di bromobutane gives mainly:-
Elimination of bromine from 2-bromobutane results in the formation of ______.
The conversion of an alkyl halide into an alkene by alcoholic KOH is classified as ______.
Reaction of trans-2-phenyl-1-bromocyclopentane with alcoholic KOH produces.
Which of the following alkyl halides will undergo SN1 reaction most readily?
An SN1 reaction at the asymmetric carbon of an enantiomerically pure chiral alkyl halide gives a product ______.
Major products A and B formed in the following reaction sequence are: