Advertisements
Advertisements
Questions
How will you bring about the following conversion?
Toluene to benzyl alcohol
How the following conversion can be carried out?
Toluene to benzyl alcohol
How will you bring about the following conversion in not more than two steps?
Toluene to Benzyl alcohol
Advertisements
Solution 1
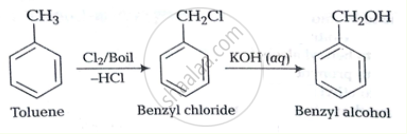
Solution 2
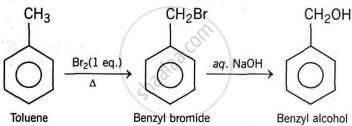
RELATED QUESTIONS
Discuss the mechanism of alkaline hydrolysis of bromomethane.
Which would undergo SN2 reaction faster in the following pair and why ?

Write the structures of A, B and C in the following:

Write the isomers of the compound having the formula C4H9Br.
Write the structure of the major organic product in the following reaction:
\[\ce{CH3CH2CH2OH + SOCl2 ->}\]
What happens when ethyl chloride is treated with aqueous KOH?
Given reasons: SN1 reactions are accompanied by racemization in optically active alkyl halides.
Identify 'A' in the following reaction -

(a) 2- Bromo-2 methylbutane
(b) 1 -Bromo-2,2-dimethylpropane
(c) 1 - Bromo - 3 -methylbutane
(d) 1 - Bromo- 2 -methylpropane
What is the action of the following on ethyl bromide?
silver acetate
Answer the following question.
Write one stereochemical difference between SN1 and SN2 reactions.
In the SN1 reaction, racemization takes place. It is due to:
The order of reactivities of the following alkyl halides for an SN2 reaction is:
Optically active isomers but not mirror images are called ____________.
An organic molecule necessarily shows optical activity if it ____________.
Which of the following is a chiral compound?
The reaction of C6H5–CH=CH–CH3 with HBr produces:
Which of the following alkyl halides will undergo SN1 reaction most readily?
Ethylene chloride and ethylidene chloride are isomers. Identify the correct statements.
(i) Both the compounds form same product on treatment with alcoholic KOH.
(ii) Both the compounds form same product on treatment with aq.NaOH.
(iii) Both the compounds form same product on reduction.
(iv) Both the compounds are optically active.
The number of chiral carbons present in the molecule given below is ______.

Which of the following reactions is an example of nucleophilic substitution reaction?
