Advertisements
Advertisements
Question
Which of the following alkyl halides will undergo SN1 reaction most readily?
Options
\[\ce{(CH3)3C-F}\]
\[\ce{(CH3)3C-Cl}\]
\[\ce{(CH3)3C-Br}\]
\[\ce{(CH3)3C-I}\]
Advertisements
Solution
\[\ce{(CH3)3C-I}\]
Explanation:
\[\ce{(CH3)3C-I}\] will undergo SN1 reaction most readily as C-I bond is weakest, due to the large difference in the size of carbon and iodine.
APPEARS IN
RELATED QUESTIONS
Discuss the mechanism of alkaline hydrolysis of bromomethane.
Write the major products(s) in the following:

In the following pair of halogen compounds, which compound undergoes a faster SN1 reaction?

How will you bring about the following conversion?
Toluene to benzyl alcohol
What happens when chlorobenzene is subjected to hydrolysis?
What is the action of the following on ethyl bromide:
moist silver oxide
What is the action of the following on ethyl bromide?
moist silver oxide
The order of reactivity of the given haloalkanes towards nucleophile is:
SN2 mechanism proceeds through intervention of ____________.
Which of the following is the correct order of decreasing SN2 reactivity?
Which of the following compounds is optically active?
2-Bromopentane is heated with potassium ethoxide in ethanol. The major product obtained is ____________.
Which reagent will you use for the following reaction?
\[\ce{CH3CH2CH2CH3 -> CH3CH2CH2CH2Cl + CH3CH2CHClCH3}\]

Which of the statements are correct about above reaction?
(i) (a) and (e) both are nucleophiles.
(ii) In (c) carbon atom is sp3 hybridised.
(iii) In (c) carbon atom is sp2 hybridised.
(iv) (a) and (e) both are electrophiles.
Which one is the correct order of nucleophilic strength (pKa) of following nucleophiles?
Racemisation occurs in ______.
Give the mechanism of the following reaction:
\[\ce{CH3CH2OH ->[H2SO4][413 K] CH3CH2-O-CH2CH3 + H2O}\]
Which alkyl halide from the following pair would you expect to react more rapidly by an SN2 mechanism? Explain your answer.
\[\begin{array}{cc}\ce{CH3CH2CHCH3}\\
\phantom{.....}|\\
\phantom{......}\ce{Br}\\
\end{array}\] or \[\begin{array}{cc}\phantom{.......}\ce{CH3}\\
\phantom{...}|\\
\ce{H3C - C - Br}\\
\phantom{...}|\\
\phantom{.......}\ce{CH3}\\
\end{array}\]
The correct order of increasing reactivity of
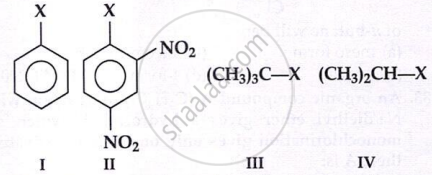
C-X bond towards nucleophile in the following compounds is:
Which of the following reactions is an example of nucleophilic substitution reaction?
