Advertisements
Advertisements
Question
Write the structures of A, B and C in the following:

Advertisements
Solution
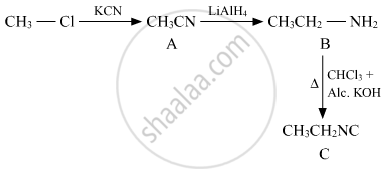
APPEARS IN
RELATED QUESTIONS
Out of  , which is more reactive towards SN1 reaction and why?
, which is more reactive towards SN1 reaction and why?
Which alkyl halide from the following pair would you expect to react more rapidly by an SN2 mechanism? Explain your answer.
CH3CH2CH2CH2Br or \[\begin{array}{cc}
\ce{CH3CH2CHCH3}\\
\phantom{...}|\\
\phantom{....}\ce{Br}\
\end{array}\]
Which of the following is optically inactive?
Most reactive halide towards SN1 reaction is ____________.
Which one is the correct order of nucleophilic strength (pKa) of following nucleophiles?
Give reason for the following:
The product formed during SN1 reaction is a racemic mixture.
Optical activity of an enantiomeric mixture is +12.6° and the specific rotation of (+) isomer is +30°. The optical purity is ______ %.
Discuss SN2 mechanism of methyl bromide using aqueous KOH.
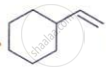 HCI, Major product ______.
HCI, Major product ______.
Which of the following is halogen exchange reaction?
