Advertisements
Advertisements
Question
Give the structure and name of the product in the following reaction.
Sulphonation of chlorobenzene.
Chemical Equations/Structures
Advertisements
Solution
When chlorobenzene is treated with concentrated sulphuric acid (H2SO4) or fuming sulphuric acid (oleum, H2S2O7), it undergoes electrophilic substitution at the aromatic ring. The sulphonation occurs at the para position relative to the chlorine atom due to its electron-withdrawing yet ortho/para-directing nature.
- Reaction equation:
\[\ce{C6H5Cl + H2SO4 ->[conc. H2SO4, Δ][]o-ClC6H4SO3H + p-ClC6H4SO3H}\]
(Chlorobenzene) + (sulphonating agent) → ortho- and para-chlorobenzenesulphonic acids - Structure of the Product: p-Chlorobenzenesulphonic acid
- The chlorine is attached to one carbon of the benzene ring.
- The –SO3H (sulphonic acid group) is attached to the para-position relative to Cl.
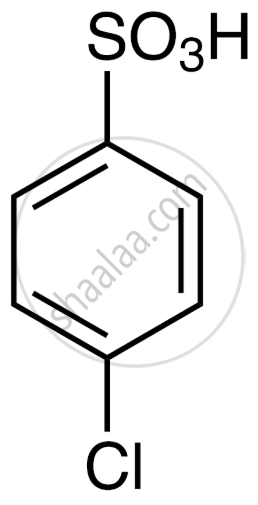
- Name of the Product: para-Chlorobenzenesulphonic acid (or 4-Chlorobenzenesulphonic acid)
shaalaa.com
Is there an error in this question or solution?
Chapter 6: Haloalkanes and Haloarenes - REVIEW EXERCISES [Page 604]
