Advertisements
Advertisements
Question
Name the following halide according to the IUPAC system and classify it as an alkyl, allyl, benzoyl (primary, secondary, tertiary), vinyl or aryl halide:
p-ClC6H4CH2CH(CH3)2
Advertisements
Solution
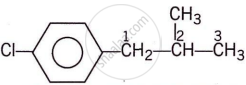
IUPAC Name: 1-(4-chlorophenyl)-2-methylpropane
Classification: Aryl halide
APPEARS IN
RELATED QUESTIONS
Draw the structure of the major monohalo product in the following reaction:

Name the following halide according to the IUPAC system and classify it as an alkyl, allyl, benzoyl (primary, secondary, tertiary), vinyl or aryl halide:
(CH3)3CCH2CH(Br)C6H5
Name the following halide according to the IUPAC system and classify it as an alkyl, allyl, benzoyl (primary, secondary, tertiary), vinyl or aryl halide:
CH3CH(CH3)CH(Br)CH3
Name the following halide according to the IUPAC system and classify it as an alkyl, allyl, benzoyl (primary, secondary, tertiary), vinyl or aryl halide:
CH3C(Cl)(C2H5)CH2CH3
Which is the correct IUPAC name for \[\begin{array}{cc}
\ce{CH3 - CH - CH2 - Br}\\
\phantom{}|\phantom{.......}\\
\phantom{}\ce{C2H5}\phantom{....}
\end{array}\]?
The reaction of toluene with chlorine in the presence of iron and in the absence of light yields ______.
Which of the carbon atoms present in the molecule given below are asymmetric?
\[\begin{array}{cc}
\phantom{}\ce{HO}\phantom{.....}\ce{OH}\phantom{..}\ce{H}\phantom{.....}\ce{O}\phantom{..}\\
\phantom{..}\backslash\phantom{.....}|\phantom{....}|\phantom{.....}//\phantom{.}\\
\ce{\overset{a}{C} - \overset{b}{C} - \overset{c}{C} - \overset{d}{C}}\\
\phantom{..}//\phantom{.....}|\phantom{....}|\phantom{....}\phantom{.}\backslash\phantom{...}\\
\phantom{}\ce{O}\phantom{......}\ce{H}\phantom{...}\ce{OH}\phantom{...}\ce{H}\phantom{}\\
\end{array}\]
Name the following halide according to IUPAC system and classify it as alkyl, allyl, benzyl (primary, secondary, tertiary), vinyl or aryl halide:
\[\ce{CH2CH2C(CH3)2CH2I}\]
Name the following halides according to the IUPAC system and classify them as alkyl, allyl, benzyl (primary, secondary, tertiary), vinyl or aryl halides:
CH3C(C2H5 )2CH2Br
Name the following halide according to IUPAC system and classify them as alkyl, allyl, benzyl (primary, secondary, tertiary), vinyl or aryl halide:
CH3C(Cl)(C2H5)CH2CH3
Name the following halide according to the IUPAC system and classify it as alkyl, allyl, benzyl (primary, secondary, tertiary), vinyl, or aryl halide:
\[\ce{CH3C(C2H5)2CH2Br}\]
Write the structure of the following organic halogen compound.
1,4-Dibromobut-2-ene
Name the following halides according to IUPAC system and classify them as alkyl, allyl, benzyl (primary, secondary, tertiary), vinyl or aryl halides:
CH3C(Cl)(C2H5 )CH2CH3
Name the following halide according to IUPAC system and classify them as alkyl, allyl, benzyl (primary, secondary, tertiary), vinyl or aryl halides:
\[\ce{CH3C(Cl)(C2H5)CH2CH3}\]
Name the following halide according to IUPAC system and classify it as alkyl, allyl, benzyl (primary, secondary, tertiary), vinyl or aryl halides:
\[\ce{CH3C(Cl)(C2H5)CH2CH3}\]
Name the following halide according to IUPAC system and classify as alkyl, allyl, benzyl (primary, secondary, tertiary), vinyl or aryl halides:
\[\ce{CH3C(C2H5)2CH2Br}\]
Name the following halide according to IUPAC system and classify them as alkyl, allyl, benzyl (primary, secondary, tertiary), vinyl or aryl halides:
\[\ce{CH3C(C2H5)2CH2Br}\]
IUPAC name of \[\begin{array}{cc}
\phantom{}\ce{CH3}\phantom{..........}\ce{Br}\phantom{........}\\
\phantom{}|\phantom{................}|\phantom{.........}\\
\phantom{}\ce{CH3CHCH2CHCHCH2CH3}\phantom{}\\
\phantom{}|\phantom{..}\\
\phantom{..}\ce{CH3}\phantom{}
\end{array}\] is ______.
Which is the correct IUPAC name for 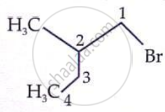 is______.
is______.
